Category: Medical Ethics

Big Trouble in Little Pharma
The Excruciating Inspection of David Geier’s DAP Pharmaceuticals

Pig Heart Xenografts for Infants
Organ transplant is a potentially lifesaving medical intervention, but there is a critical lack of donor organs. Even in a wealthy country like the US, there are about 100,000 people on the waiting list for an organ transplant, but only about 23 thousand organs become available each year. About 6 thousand people die each year while on the waiting list. For this...

Old Doctor Yells At Clouds
I’m at that age, 67 going in 68, where it is reasonable to yell at the clouds. Which, come to think of it, may be a good title for another collection of my SBM essays. Aging does indeed suck, but it is, usually, better than the alternative. As we, and by we I mean my family and me, age we get the...
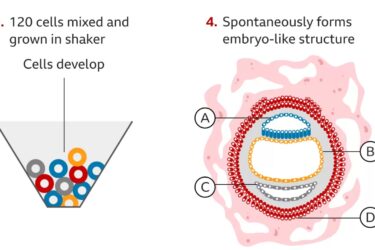
Complete Human Embryo Model Made From Stem Cells
Researchers create an embryo model from human embryonic stem cells.

Examining COVID-19 misinformation propagated by US physicians
A new paper documents COVID-19 medical misinformation shared by US physicians on social media

COVID-19 vaccines and the Nuremberg Code
Antivaxxers love to claim that vaccine mandates (especially COVID-19 vaccine mandates) violate the Nuremberg Code and call for Nuremberg-style tribunals to hold public health and vaccine advocates "accountable". As usual, they have no idea what they are talking about. This is also not a new antivax narrative, although what is unprecedented is that what was once fringe even among antivaxxers is now...
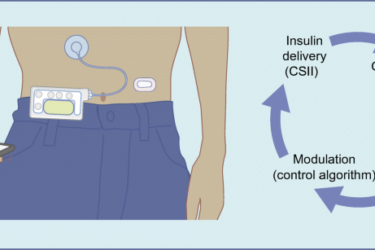
DIY Diabetes Treatment
Can DIY medicine work?

About those “19 Errors,” Part Two
A critical and evidence-based response to the alleged errors journalist Jesse Singal found in my guest post reviewing Abigail Shrier's book on trans youth. The second of two parts.

About those “19 errors,” part one
Journalist Jesse Singal took issue with Dr. Rose Lovell and Dr. AJ Eckert's guest posts about Abigail Shrier's book on transgender teens, Dr. Harriet Hall's review of the book, and the medical care of these teens, claiming that the posts contained "19 errors." In this post, Dr. Lovell responds to her share of these "19 errors," most of which are not errors.

Abigail Shrier’s Irreversible Damage: A Wealth of Irreversible Misinformation
A critical, science-based analysis of Abigail Shrier's book Irreversible Damage.

