Category: Politics and Regulation
Acupuncture for Heart Attacks and more State-Sanctioned Pseudoscience
States default to a private organization run by acupuncturists and TCM practitioners to vet continuing education courses, with predictable results.

The Lysenko-ization of federal science takes a big step forward
From the beginning, Trump science policy has been Lysenko 2.0, in which ideology, not scientific promise and rigor, dictates federal grantmaking. OMB Director Russell Vought's proposed rules de-emphasizing peer review and placing political appointees in charge of final grantmaking decisions do Lysenko proud.

How Unbekoming! The central delusion of MAHA
What do vitamin K shots, vaccines, MTHFR variants, and statins have in common? The answer is what I call the central delusion behind MAHA, which was inadvertently revealed by a blogger with the 'nym Unbekoming.

MAHA vs. the FDA: Dredging up old anti-regulation revisionist history
Recently, I've noticed articles from outlets aligned with MAHA calling for the elimination of the FDA. It's all recycled "health freedom" revisionist history and ahistorical nonsense.

SCOTUS conversion therapy decision “opens a dangerous can of worms”
“We are on a slippery slope now: For the first time, the Supreme Court has interpreted the First Amendment to bless a risk of therapeutic harm to children by limiting the State’s ability to regulate medical providers who treat patients with speech.” Justice Jackson, dissenting
KevinMD platforms criticism of MAHA by a “functional medicine” practitioner
Although some of the criticisms of Robert F. Kennedy Jr.'s MAHA movement are spot on, why did KevinMD platform a practitioner of functional medicine woo to make them?

MAHA and science-based public health: Can’t we all just get along?
The answer is: Very likely not, at least not as long as MAHA embraces quackery and antivax pseudoscience—not that that didn't stop STAT News from ignoring the elephant in the room, vaccines, in search of a "kumbaya" moment between MAHA and public health.
Legislative Alchemy: Licensing reflexologists and other practitioners of pseudoscience
State legislatures are considering bills that would legitimize pseudoscience like reflexology and reiki by recognizing their practitioners as health care professionals.
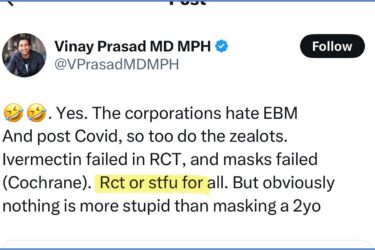
Dr. Vinay Prasad Said He Would Deliver New COVID Vaccine RCTs. He Failed and Should STFU.
It turns out that Tweeting about RCTs 100+ times is a lot easier than delivering even a single RCT.


Can Medical Schools Really Teach 71 Nutrition ‘Competencies’? Should They?
In recent years, nutrition has become the focus of renewed attention in medical education. Advocates argue that physicians receive too little formal training about diet and that more comprehensive nutrition education is needed to address chronic diseases such as obesity, diabetes, cardiovascular disease, and certain cancers. A greater focus on the “root causes of chronic diseases” is a mantra of the Make...