Tag: Food and Drug Administration

MAHA vs. the FDA: Dredging up old anti-regulation revisionist history
Recently, I've noticed articles from outlets aligned with MAHA calling for the elimination of the FDA. It's all recycled "health freedom" revisionist history and ahistorical nonsense.
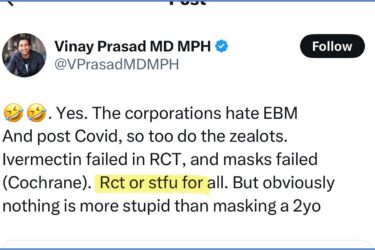
Dr. Vinay Prasad Said He Would Deliver New COVID Vaccine RCTs. He Failed and Should STFU.
It turns out that Tweeting about RCTs 100+ times is a lot easier than delivering even a single RCT.

One year of Robert F. Kennedy Jr. as HHS Secretary: The Lancet reacts, and so do I
Editors of The Lancet published an op-ed decrying RFK Jr.'s one year of failure at HHS. They're correct about RFK Jr, but they are not blameless in what has happened.

RFK Jr. is definitely coming for your vaccines (part 6): Liability, ethics, and policy-based evidence making
As 2026 dawns, look for HHS Secretary Robert F. Kennedy Jr. to ramp up his assault on vaccines using policy-based evidence making and altering Vaccine Court standards.

The FDA under MAHA control: Weakening the quack Miranda warning on supplements
The FDA sent a letter to the supplement industry assuring that it would make it easier for them to hide disclaimers (which we like to call "quack Miranda warnings") about unproven health claims for supplements. What does this tell us about MAHA?

“Best practices”: The buzzword that the Trump administration will use to eliminate as many vaccines as RFK Jr. can
The CDC Advisory Committee on Immunization Practices met last week, eliminating the universal recommendation for the birth dose of the hepatitis B vaccine. There was, however, a more insidious threat to vaccines in a White House press release after the meeting cloaked under the buzzword "best practices."

Robert F. Kennedy, Jr. is definitely coming for your vaccines, part 5: VAERS and a useful idiot at the FDA
In the sadly continuing story of how Health and Human Services Secretary RFK Jr. is working to eliminate vaccines, we look at a useful idiot, Dr. Vinay Prasad, and how he is weaponizing dead children to justify making vaccines harder to approve.

Read the Comments: “Just Do the Opposite of Whatever This Administration Recommends!”
It's obviously not good that people have lost trust in the FDA, NIH, and CDC. However, since those once vulnerable institutions are now lead by naked emperors, it's good that the American public, at least those that read the news, has uniformly recognized they have no clothes.

Robert F. Kennedy, Jr. is definitely coming for your vaccines, part 2: VAERS, the FDA, and ACIP
The Food and Drug Administration appears to be about to weaponize a classic antivax trope against COVID-19 vaccines, the abuse of reports to the Vaccine Adverse Events Reporting System to falsely portray them as deadly, even as news reports suggest that RFK Jr. will further restrict access to COVID-19 and three childhood vaccines. Here we go again.