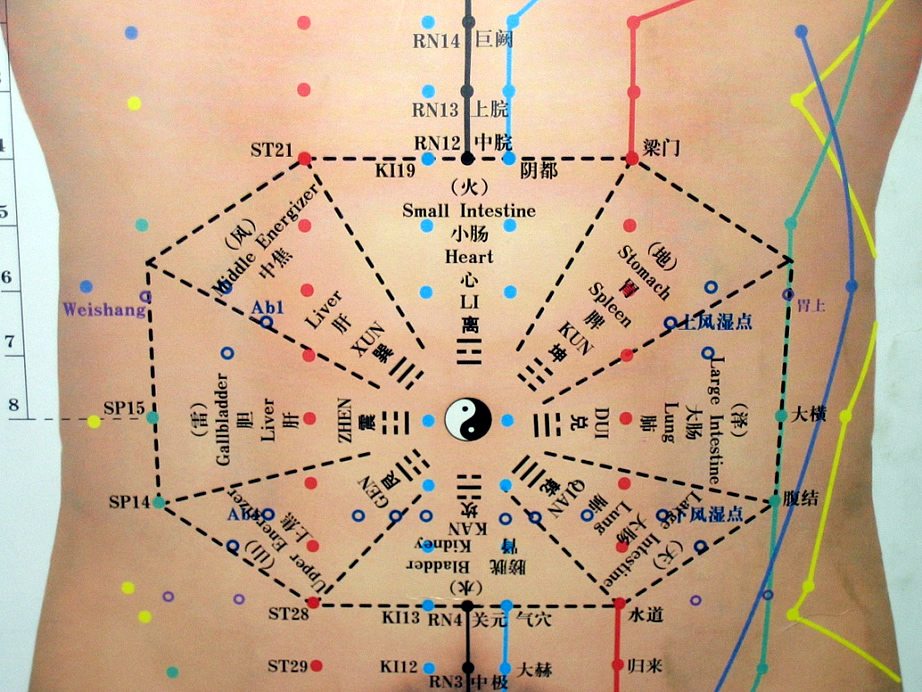
Acupuncture for Heart Attacks and more State-Sanctioned Pseudoscience
States default to a private organization run by acupuncturists and TCM practitioners to vet continuing education courses, with predictable results.
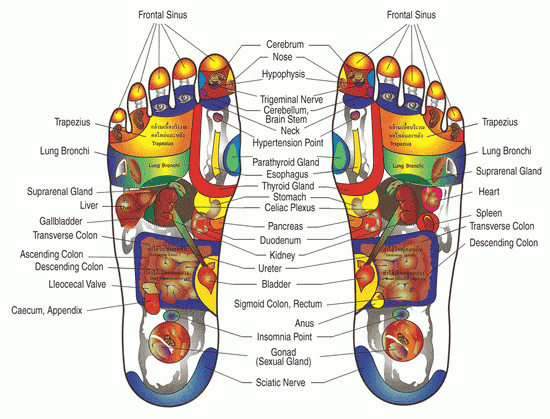
Legislative Alchemy: Licensing reflexologists and other practitioners of pseudoscience
State legislatures are considering bills that would legitimize pseudoscience like reflexology and reiki by recognizing their practitioners as health care professionals.

Legislative Alchemy: “Naturopathic Doctor” licensing is bad medicine for Florida
Bad medicine, bad laws, bad choice
Federal employment rights agency inundated with thousands of COVID-related discrimination claims
Thousands of workers have filed complaints with the EEOC alleging COVID-related employment discrimination. It may take years of litigation to sort out the application of federal anti-bias laws to these claims.
State Attorneys General pursue consumer protection law claims against stem cell clinics
State Attorneys General are pursuing stem cell clinics offering unproven therapies and engaging in fake clinical trials using state consumer protection and false advertising laws, seeking monetary penalties and injunctive relief. Until there is rational, comprehensive stem cell regulation, these actions can help fill the regulatory gap.
Good faith doctoring or greedy drug dealing? SCOTUS hears opioid prescribing cases
Physicians running opioid "pill mills" were convicted of violating the Controlled Substances Act and given substantial prison sentences. The U.S. Supreme Court will soon decide whether they were entitled to a "good faith" defense at trial.

FTC targets physicians’ COVID claims with cease-and-desist letters
The FTC recently announced it had issued 20 more cease-and-desist demands to physicians and others claiming their products and services prevent or treat COVID-19 without sufficient scientific backing. Unfortunately, this has not stopped many of the targets from making other bogus health claims.

State legislators dispense with standard of care for COVID treatment and encourage medical misinformation
State legislatures are considering bills that protect doctors and other health care providers from being held accountable for using unproven COVID-19 treatments and spreading medical misinformation.



SCOTUS conversion therapy decision “opens a dangerous can of worms”
“We are on a slippery slope now: For the first time, the Supreme Court has interpreted the First Amendment to bless a risk of therapeutic harm to children by limiting the State’s ability to regulate medical providers who treat patients with speech.” Justice Jackson, dissenting