Category: Medical devices

Signos Sells a Continuous Glucose Monitor, But Not to Diabetics
Signos is asking customers to pay for the privilege of testing their glucose monitoring system.

Plenity – A New Weight Loss Pill
Plenity is a new weight loss pill designed to create a sense of fullness. It is backed by a single study where users had an average weight loss of 22 pounds. Not an effective way to achieve ideal weight, but may help some people when combined with diet and exercise.
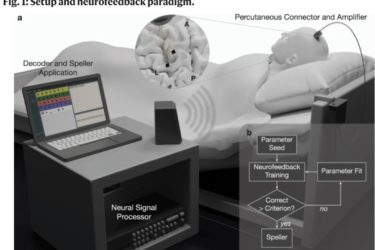
Paralyzed Patient Communicates with Brain Implant
Brain-machine interfaces continue to advance.
Socks to Treat High Blood Pressure?
Socks that lower blood pressure? The claims for Boliav socks are too incoherent to make sense of. I can't take them seriously.

This Scary Mask Is Not for Halloween: It’s for Anti-Aging Skin Care
Evidence is lacking for CurrentBody's "anti-aging" LED mask.

Innovo for Bladder Leakage
An expensive pair of shorts promises to cure bladder leaks by using electrical stimulation to produce pelvic floor contractions, essentially doing the Kegel exercises for you. The science is insufficient and the marketing is misleading.

The Blu Room
Blu Rooms are an expensive way to relax. The testimonials and the medical history of the inventor are not believable. No science, but good for a laugh.

Bioptron: Too Silly to Write about
Bioptron is a silly device claiming to work through unproven means, but basically seems to be an extremely colorful flashlight.

Radioactive 5G Pendants
Authorities had to warn the public not to use radioactive products to protect against harmless 5G.
Nuubu: Here We Go Again! Recycling Debunked Foot Detox Myths
Companies come and go, but the claims remain the same, that you can (insert claim) with (insert product) without any evidence. A new company offering magical footpads are just putting new wine in old bottles.

