Tag: Alzheimer’s disease
New Review Casts Doubt On Alzheimers Drugs But Is Controversial
There's a lot less than meets the eye to a recent Cochrane review that casts doubt on the efficacy of drugs for Alzheimer's disease.

Prions. Why did it have to be prions? (Again.)
Prions. Why did it have to be prions? (Again.) The antivax trope that vaccines cause prion disease is an old one, and antivaxxers are trying desperately to resurrect it to apply to COVID-19 vaccines.

Adios Aduhelm
The controversial and never-proven-effective drug to treat Alzheimer's disease, aducanumab (Aduhelm) has been discontinued.

FDA Approves Leqembi for Alzheimer’s
FDA upgrades Leqembi for AD from accelerated to traditional approval based on latest clinical data.

AI as a Diagnostic Tool
Using AI systems for pattern recognition in early diagnosis of dementia shows the potential of this tool.

Congress criticizes the FDA and the Aduhelm approval
A Congressional report on the approval of the Alzheimer's drug Aduhelm identified serious concerns about the FDA's processes
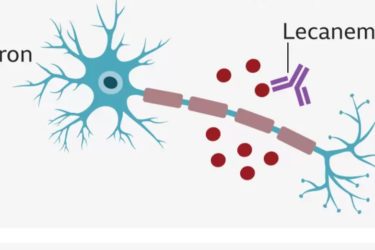
Another Controversial Alzheimer’s Drug
A new treatment for Alzheimer's Disease is promising, but we're not there yet.

Can supplements improve memory and cognition?
Can you reduce the risk of cognitive decline with dietary supplements?
Fraud, Scientific Rigor, and Alzheimer’s Research
A stunning case of possible fraud in Alzheimer's research reinforces the need for scientific rigor at every level.
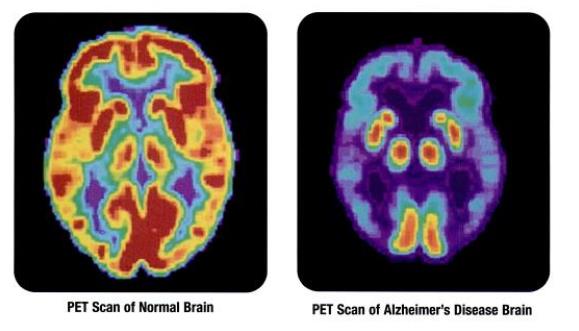
COVID Associated With Neurodegenerative Disorders
COVID infection has been associated with an increased risk of neurodegenerative and neurovascular disease - another reason to keep our eye on this serious illness.

