Month: August 2020
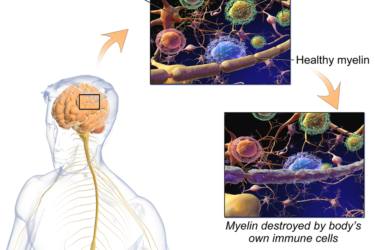
The First Multiple Sclerosis Patient
Twenty years before Charcot described the nerve-destroying disease multiple sclerosis, an illegitimate British noble spent much of his adult life describing the disease and its effects.
Trump administration announces some COVID-19 tests can skip FDA review, providing new opportunities for dubious lab tests
The Trump administration unexpectedly announced that the FDA will no longer regulate some lab tests, including those for COVID-19. In addition to potentially allowing unreliable COVID tests on the market, the decision creates an opening for more bogus CAM tests.
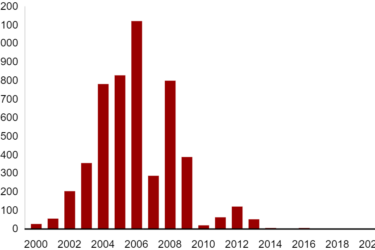
Polio Eliminated From Africa
Wild-type polio has been eliminated from Africa. Now only Pakistan and Afghanistan remain.

“(Un)Well:” Netflix’s Documentary Series Is Poor Journalism That Neglects Science
The (Un)Well documentary series on Netflix asks "Wellness: does it bring health and healing, or are we falling victim to false promises?" But instead of answers, it offers false balance and confusion.

Does convalescent plasma work against COVID-19? Who knows?
Last night, the FDA issued an emergency use authorization (EUA) for convalescent plasma to treat COVID-19, even though there are no randomized clinical trials demonstrating efficacy and safety. Does this plasma work? Who knows? But that didn't stop the FDA from issuing the EUA, almost certainly as a result of intense political pressure from the Trump Administration.
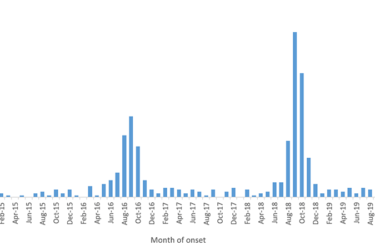
CDC Issues Warning: Be on the Lookout for a Surge in Pediatric Paralysis Cases This Fall
The CDC is warning parents and pediatric medical professionals to be on the lookout for a potentially devastating viral disease this fall. It isn't the novel coronavirus, but the impact of the pandemic might make things worse...or better.
Dexamethasone and Hydroxychloroquine: Why Randomized Controlled Trials Matter
What does the best evidence tell us about hydroxychloroquine and dexamethasone?

Oleandra – The New COVID Snake Oil
Oleandrin is being promoted as the new COVID-19 snake oil - but it is a deadly toxin.