Tag: Food and Drug Administration

Denial of germ theory and the genetic basis of disease: Two pillars of MAHA
To understand MAHA, understand the specific science that it denies.
I am a Private Citizen Seeking to Hold My Government Accountable. Dr. Vinay Prasad, a Government Doctor, Killed My YouTube Channel.
Americans do not need our government's permission to remember the words of our public officials, and I refuse to let them be silenced or censored.

Every accusation was a confession of what they wanted to do if they were to attain power
On Friday, President Donald Trump signed an executive order that would shift power over grantmaking away from career scientists to political appointees. Lysenkoism 2.0 has been made official policy, and science in the US is in deep trouble.

EBM “Boy Wonder” Vinay Prasad is toppled
Evidence-based medicine "Boy Wonder" Dr. Vinay Prasad was fired as CBER Director after less than three months on the job. What can the kerfuffle surrounding his ouster tell us about the FDA and doctors who enabled Robert F. Kennedy, Jr.?

COVID-19 contrarian Dr. Vinay Prasad meets real world responsibility
Dr. Vinay Prasad parlayed his COVID-19 contrarianism into a high-ranking position in the FDA. Now he's facing real world responsibility, and knives are out for him. Can he survive?

After ACIP: Where Robert F. Kennedy Jr. will likely go from here in his quest to eliminate vaccines
HHS Secretary Robert F. Kennedy Jr. was on The Tucker Carlson Show last week. It got me to thinking: What's next in RFK Jr.'s crusade against vaccines after he turned ACIP into an antivax committee?

Marty Makary and Vinay Prasad embrace a “more fluid concept of evidence” at the FDA
Last Thursday during a roundtable on stem cell therapies, new FDA Commissioner Marty Makary referred to EBM levels of evidence as an artificial and dogmatic construct. Apparently Dr. David Katz's "more fluid concept of evidence" now reigns at the FDA—selectively.

Attacking vaccines through the misunderstanding of medical ethics
Since becoming HHS Secretary, RFK Jr. has been deceptively weaponizing principles of evidence-based medicine against vaccines. Will weaponizing research ethics be next? It might if the ideas of one of his advisors, James Lyons-Weiler, are any indication.
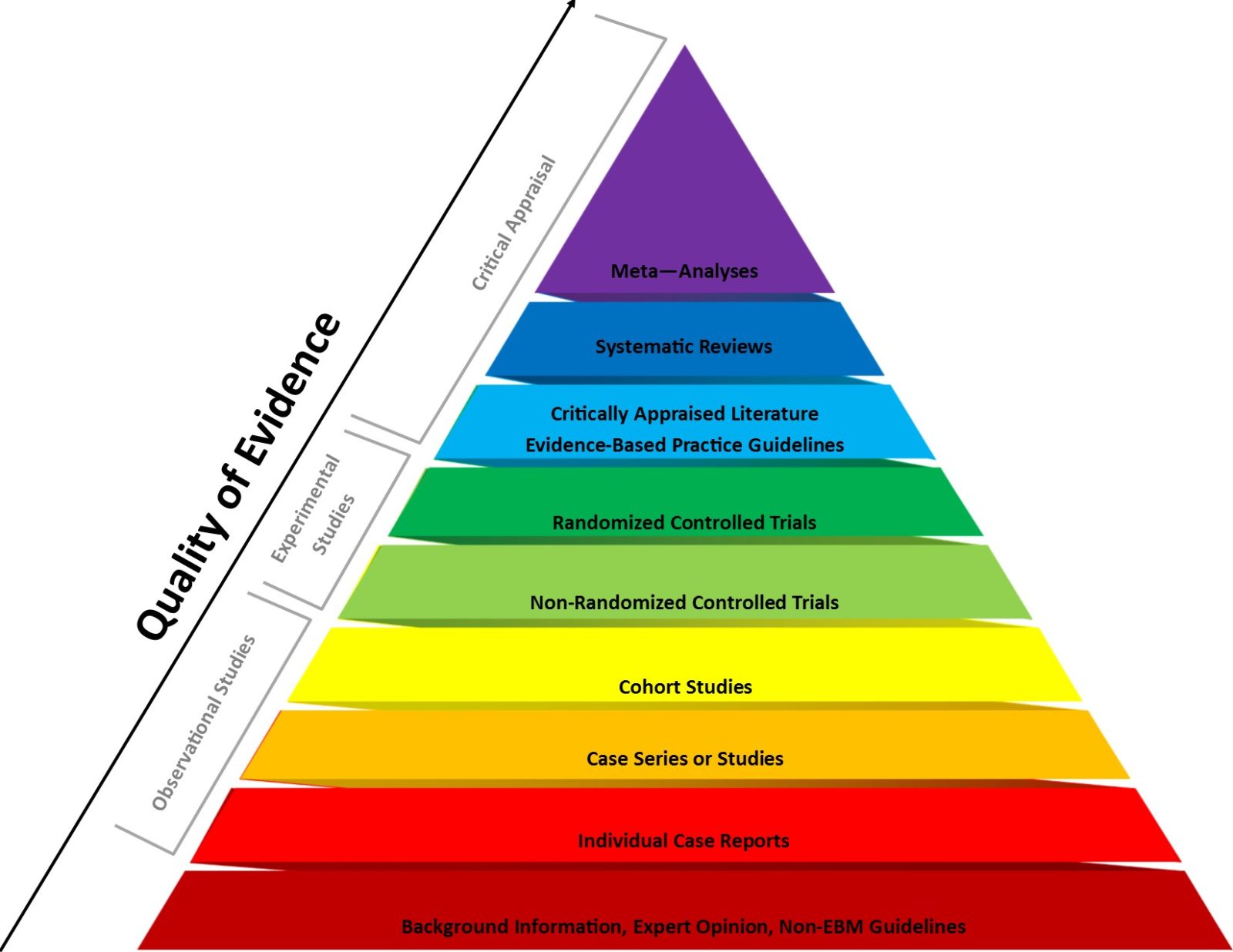
HHS is weaponizing evidence-based medicine to falsely portray vaccines as unsafe
Last week, HHS announced that all "new vaccines will undergo safety testing in placebo-controlled trials." Sounds good? Not so fast. This is really a deceptive weaponization of evidence-based medicine to undermine confidence in vaccines and eliminate at least some of them.