We spend a great deal of time in the pages of Science-Based Medicine taking down every form of pseudoscience in medicine. Of course, what we see as pseudoscience, proponents often see as emerging or cutting edge science. They are taking advantage of the fact that there is a great deal of legitimate emerging science, and they hope they can sneak past the gates by cloaking themselves in the trappings of real science (jargon, studies, their own journals, etc.). Emerging science, however, no matter how plausible and earnest, still has yet to prove itself (by definition), and has to go through the rigorous process of scientific evaluation to slowly gain acceptance. That process – sorting out what works from what doesn’t, the real from the fake – is where all the action is in SBM.
It is refreshing sometimes to talk about an emerging field that, while still experimental, is legitimate and has the potential to usher in a genuinely revolutionary treatment.
I have been following the research into brain-machine interfaces for some years, and reporting on many of the significant “baby steps” in the advance of this new technology. A recent study published in The Lancet represents another incremental and encouraging advance. Researchers at the University of Pittsburgh implanted two strips of 96 electrodes into the motor cortex of a 52 year-old woman with tetraplegia. The electrodes are capable of detecting the firing of neurons in the motor cortex and transmitting those signals to an external processor that in turn controls a fairly sophisticated robotic arm. The arm is described as having seven degrees of freedom – three dimensions of translation, three dimensions of orientation, and one dimension of grasping.
After two days the subject was able to move the robotic arm with her thoughts alone. Over the course of the 13 week study she progressively gained control of the arm and eventually was able to feed herself with the arm. While this is still very far from a “cure” for paralysis or a restoration of full function, for someone who is tetraplegic (all four limbs are paralyzed) having any independent function is a huge improvement in quality of life.
So where are we with this technology?
There are two basic approaches to such brain-machine interfaces – using scalp electrodes or using implanted brain surface electrodes. The advantage of the scalp electrodes is that they are non-invasive. The subject has to just where a cap or helmet of electrodes. There is, however, a significant disadvantage to this approach – the skull and other tissue between the brain and the electrodes significantly impedes the electrical signals from brain activity. This reduces the resolution of the information that can be read from brain activity.
Implanted electrodes have much greater resolution in terms of reading brain activity. You can have a greater number of smaller electrodes, like seeing a picture which far smaller pixels and in focus. The huge disadvantage of implanted electrodes is that they are implanted – this is an invasive procedure, with wires exiting the skull to connect to the external computer. The potential for medical complications, therefore, is far greater than for external electrodes. Research, however, seems to be favoring implanted electrodes as the resolution of scalp surface electrodes is just not sufficient.
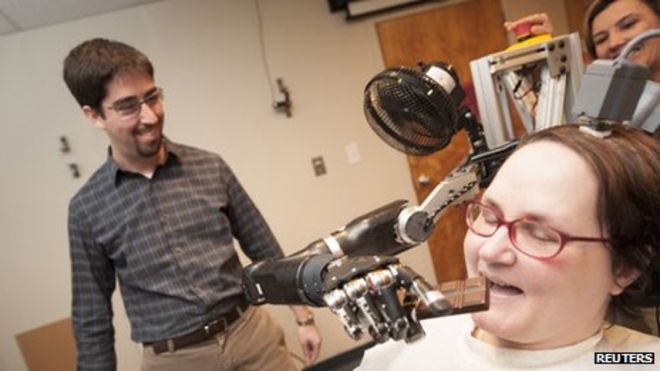
How much of a limitation this will have on the application of this technology remains to be seen. As you can see from the picture above, the subject has a device connected to the top of her head that connects to the implanted electrodes. This study took place in the lab. They hope to attach the robotic arm to her wheelchair so she can use it outside the lab, but I do wonder what the long term safety is of having implanted electrodes connected to an external device.
There are analogies in other medical technologies. Pacemakers are fully implanted electronic devices that function safely long term. Perhaps implanted electrodes could be fully internal, and communicate to the external device through blue tooth or a similar wireless technology. Then, however (like a pacemaker) the transmitter will have to be self-powered. Advances are being made on batteries and powering small implanted devices so they don’t have to be recharged, so this is plausible, but not yet available. Patient who have ports or other devices implanted that connect to the outside world are at risk of infection. This can be managed, but it does ultimately limit the life expectancy of such devices. Brain infections, however, are extremely serious, and so any such implant will have to be as safe as possible.
Perhaps a permanent brain implant with an external connector can be devised. The signal processor and any external devices could then be plugged or unplugged from the connector (in a way similar to what was portrayed in the movie, The Matrix). In any case, this is a technical hurdle that needs to be solved for such devices to be safe and effective for long term use outside the laboratory setting.
Some advances are incremental and fairly predictable – increased number of electrodes, more sophisticated computer algorithms for control, and more sophisticated robotic designs. Over the last decade these factors have been steadily improving and can be extrapolated from existing technology. It remains to be seen, however, what the limits of this extrapolation are. How many electrodes can we place on the brain for this purpose?
In summary – researchers have already proven the concept of the brain-machine interface, and the technology has already advanced to the point where functional control of a computer or a robotic device is possible. Now it really is just a matter of further refining the technology.
The next step is the machine-brain interface (and of course closing the loop with a brain-machine-brain interface). What this means is providing feedback from an external machine to the brain. This feedback essentially means artificial sensation, such as having sensors on the hand of a robotic arm that can provide feedback as to the strength of the grip. Researchers have been focusing mainly on the proof of concept of such devices, and so far the results have been very encouraging.
Last year researchers published a study in Nature involving implanted electrodes in a monkey subject. The electrodes were on the motor cortex, and the monkey was able to control a virtual robotic arm. In addition they implanted electrodes on the somatosensory cortex and the monkey was also able to learn to distinguish virtual objects by their feel.
There is further research into brain plasticity and the function of making us feel as if we are inside of, own, and control the parts of our body. These sensations, that we take for granted and may not even be aware of, are specific functions of our brains. The brain uses sensory feedback, which it coordinates with motor intention and movement, to create the sensation that we occupy our bodies. Researchers have been able to reliably trick subjects into feeling as if they occupy a virtual body simply by playing with this sensory feedback. Essentially, if you see a body or limb, and you have a physical sensation that correlates with that body being touched, in many cases that is sufficient for your brain’s processing to conclude that you are that body. Further, if that body or limb moves when and how you intend for it to move your brain will create the sensation that you own and control that body part.
What all of this means is that visual and sensory feedback can be coordinated to make the brain’s inherent wiring create the sensation that a person occupies, owns, and controls either a robotic or even a virtual body or limb. Therefore it is theoretically possible for a robotic arm controlled by implanted electrodes to feel as if it is a person’s natural arm – that it is part of them.
The existing research is very encouraging, indicating that brain-machine-brain interface technology should work well and create direct mental control of virtual or physical external devices, including robotic limbs or even an entire robotic body or exoskeleton. We do not yet know the limits of this technology, however. Brain plasticity – the ability to adapt to new inputs and functions – exists and is crucial for this technology, but it is not unlimited.
Where we are now, therefore, is right on the brink of real-world applications for patients of brain-machine interface devices. Further, by simple extrapolation of existing technology and principles, such devices should become incrementally more powerful and sophisticated. Early research is also promising for truly revolutionary applications of this technology, combined with machine-brain interface technology, to provide neuroprosthetics that can potentially replace lost or paralyzed limbs, and many other potential applications. This is one medical technology that may seem like science fiction, but is rapidly entering the realm of reality.

