Category: Health Fraud

COVID-19 antivax quacks are now “repurposing” ivermectin for cancer
A year ago, I noticed that COVID-19 quacks were touting the "repurposing" of ivermectin to treat cancer. Now, familiar COVID-19 antivaxxers—cough, cough, FLCCC—have turbocharged this quackery.

The Wellness Company: How antivaccine grift becomes plain old quackery
The Wellness Company, promoted by Dr. Peter McCullough, is the product of a trend in which antivax doctors have predictably become just quacks. At least in this case, there is an amusing quack fight at the heart of it all.

2024 Detox Trends To Watch (Out) For
Trends come and go but the popularity of detoxification and cleansing in January is eternal.

Misinformation is pervasive, and AI will turbocharge it
Is it possible to refute an infinite amount of AI-generated health misinformation?
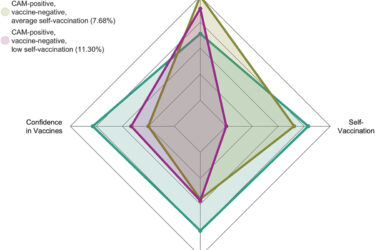
Alternative medicine and antivax: Two crappy tastes that taste crappy together—particularly when among physicians
A recent study reaffirms the high degree of correlation among physicians between antivax views and an embrace of quackery. This is an old finding that needs to be documented periodically and shows why the acceptance of non-science-based treatments by physicians endangers vaccination efforts.

Health misinformation now has powerful allies
Misinformation and conspiracy theories about health had long been a growing problem before the pandemic, but it took COVID-19 to get the government and researchers to take it seriously. Now, a new report in The Washington Post adds to previous reporting from multiple sources describing how allies of misinformation—and not just health misinformation—are striking back under the guise of defending "free speech."

Cancer quack Stanislaw Burzynski continues to prey on cancer patients in 2023
This year, cancer quack Dr. Stanislaw Burzynski turned 80. Unfortunately, he doesn't seem to be slowing down charging patients with advanced cancer huge sums for false hope.

Quoth quacks, “The medical consensus has changed before, making my quackery science!”
Brave maverick doctors (i.e., quacks) have long tried to portray themselves as "innovators" challenging an ossified medical consensus for the good of patients. This tradition continues among COVID-19 quacks, in particular the Frontline COVID-19 Critical Care Alliance and its founders.

Examining COVID-19 misinformation propagated by US physicians
A new paper documents COVID-19 medical misinformation shared by US physicians on social media

The Ohio State Medical Board has finally suspended the medical license of antivax quack Sherri Tenpenny
Last week, the Ohio State Medical Board suspended the medical license of Dr. Sherri Tenpenny, a longtime antivax quack. The only question is: What took them so long, and why did it take the pandemic for them to act? Also, is there less to this action than meets the eye?

