I’ve been saying for a while that I needed to step back from all the madness being inflicted upon US public health, medicine, and biomedical research by Secretary of Health and Human Services Robert F. Kennedy, Jr. under the guise of his “make America healthy again” (MAHA) movement, and last week I intended to to just that with a new study published a couple of weeks ago in JAMA Network Open on outcomes in breast cancer patients who used “complementary and alternative medicine” (CAM); that is, until I got distracted by the appearance of a respected oncologist and cancer center director at the MAHA Institute‘s “Massive Epidemic of Vaccine Injury” (MEVI) Roundtable. I had thought about doing a followup post on the content of the conference itself, but, really, it was a week ago, which in these times seems like an eternity. Suffice to say before moving on to today’s topic that the MEVI Roundtable was basically as bad as I had predicted, so full of a mixture of old long-debunked antivax talking points and newer COVID-19 vaccine-era equally debunked talking points that it basically reminded me of Autism One conferences, c.2011, only without the MMS (bleach) treatments for autism—and I suspect that’s just because the new generation of antivaxxers hasn’t embraced bleach yet. One statement by antivax pediatrician Larry Palevsky should tell you all you need to know about the conference, when he described all vaccinated children as “injured” and stated adamantly that he never gives vaccines. Also, antivax activist Del Bigtree was a featured speaker. ‘Nuff said.
But back to the study, Use of Complementary and Alternative Medicine in the Management of Breast Cancer, whose corresponding author is Daniel J. Boffa, MD, MBA, Division of Thoracic Surgery, Yale School of Medicine; so a shoutout to Steve Novella’s former university before he retired last summer. Conveniently, since I myself am a breast cancer surgeon, researcher, and expert, it’s the perfect sort of topic for me. Also, I can’t help but note that just last week, HHS held its National Conference on Women’s Health, which, in addition to the usual sorts of topics relevant to women’s health (e.g., cervical and breast cancer, infertility, and menopause) included a fair amount of “CAM” material with a focus on Lyme disease (for no apparent reason), and talks by functional medicine doctors like Eboni Cornish (of Amen Clinics) and Tammy Born Huizenga, of the Born Clinic, which offers every “integrative” quackery under the sun, including chelation therapy, IV vitamins, heavy metal testing, and ozone therapy. Let’s just say that MAHA is almost as much about “integrating” quackery with oncology (and medicine in general) as many of the “integrative medicine” activists that I used to write about on a regular basis.
Let’s take a look at the study.
CAM (integrative medicine) vs oncology
I will say one thing right off the bat about the study is that the authors use outdated terminology to describe the “integration” of quackery with oncology, specifically the old term “complementary and alternative medicine” (CAM), rather than “integrative medicine. I could go on and on about the evolution of terminology in which “alternative medicine” became “complementary and alternative medicine,” which ultimately became “integrative medicine,” not because the new terms illuminated or clarified, but rather because they obfuscated what “integrative” doctors (and oncologists) have been trying to do since the 1990s, at least. Basically, “CAM” and “integrative medicine” are branding terms, not real specialties. On second thought, maybe it’s better that the authors used an older term that includes “alternative medicine” in it, given that “integrative” medicine specialists have desperately wanted to eliminate anything with a hint of “alternative” in it.
As for the study itself, it’s a fairly straightforward retrospective cohort study that examined over 2 million women with breast cancer whose cases were tracked in the National Cancer Database (NCDB). The NCDB is a database sourced from hospital registry data collected in more than 1,500 facilities accredited by the American College of Surgeons Commission on Cancer (CoC). It captures approximately 70% of newly diagnosed cancers in the US and has long served as a very important database for clinical cancer research and outcomes research, as well as to establish quality benchmarks for cancer care in the US that serve as the basis of quality improvement projects. As the study describes it:
The NCDB is a database of oncology patients developed through a collaboration between the Commission on Cancer of the American College of Surgeons and the American Cancer Society.19 The NCDB Participant User File for 2022, which contains deidentified patient information, was used for the purpose of this study. Female patients who were diagnosed with breast cancer from 2011 through 2021 were included. The 11 983 patients who were missing treatment information or who were missing values for traditional therapy and/or CAM were excluded. A sensitivity analysis did not identify any obviously important differences between this group and the study population. The study was approved by the Yale University Institutional Review Board, and patient consent was waived due to use of deidentified data. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guideline was followed.
One issue—whether it’s a problem or not is debatable—that I will note right away is exactly how “CAM” is defined in the NCDB:
Complementary or alternative treatment, defined in the NCDB as treatment administered by nonmedical personnel.
However, one has to do the study with the dataset that one has available, so to speak, and that’s what the investigators at Yale did.
Definition aside, with the rise of “integrative oncology” and, as I like to call CAM/integrative medicine in academic medical centers, “quackademic medicine,” sadly a lot of “integrative therapies” these days are administered by real, bona fide MDs and DOs, some even board-certified in oncology. (One might remember times that I’ve written about the Society for Integrative Oncology and how “integrative oncology” has been uncritically promoted at large oncology meetings like the American Society of Clinical Oncology, to the point where ASCO even endorsed SIO guidelines for breast cancer and partnered with SIO to develop more guidelines (e.g., for cancer pain), apparently regardless of how quacky they are, and Nature Reviews Cancer has published at least one uncritical review touting “integrative oncology.) Too bad this is yet another study that suggests not just that “integration” of CAM into oncology doesn’t help patients but that it probably actively harms them.
To look for differences in outcomes, the authors examined the cohort of women with breast cancer and then adjusted for relevant variables known to affect outcomes of breast cancer treatment, such as age, cancer stage at diagnosis, insurance status, region, race, Charlson comorbidity index (a standardized index of how “healthy” the patient was at the time of diagnosis), and facility where treated (private, academic, or unknown). They then compared four groups:
- “Traditional” therapy (science-based oncology)
- CAM plus “traditional”
- CAM alone
- No treatment
First, here’s the graph of unadjusted overall survival:
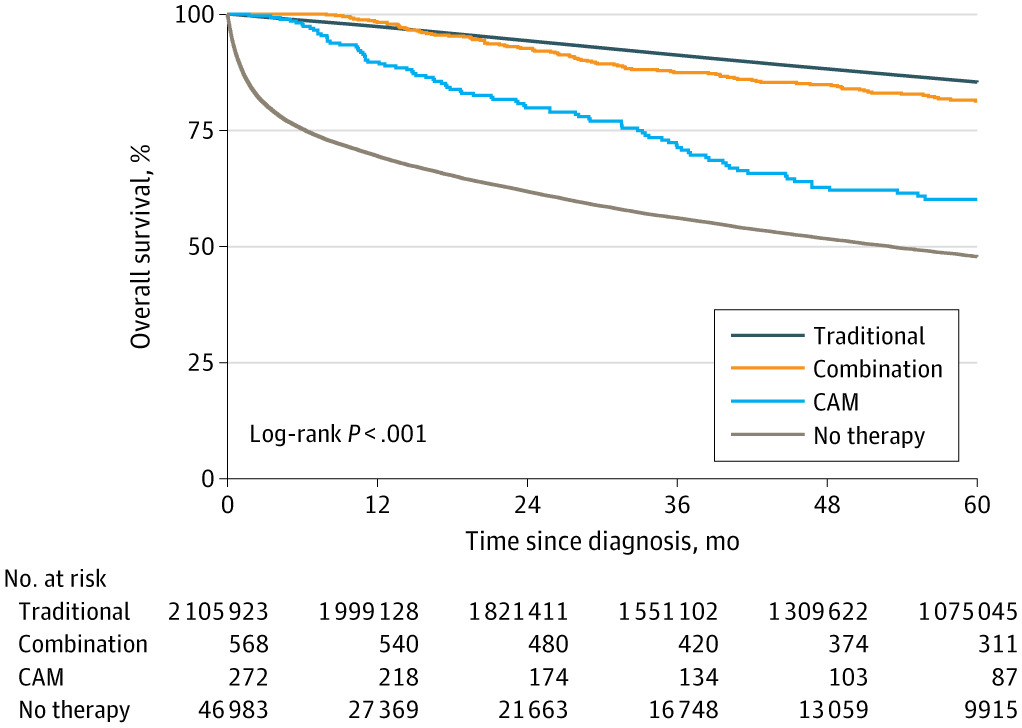
The trend is clear, but from this graph you might think there’s no real difference between using SBM and using SBM plus CAM. However, when the authors applied an adjusted Cox analysis, using patients receiving exclusively traditional therapy as the reference of 1.0, then the hazard ratios for mortality were:
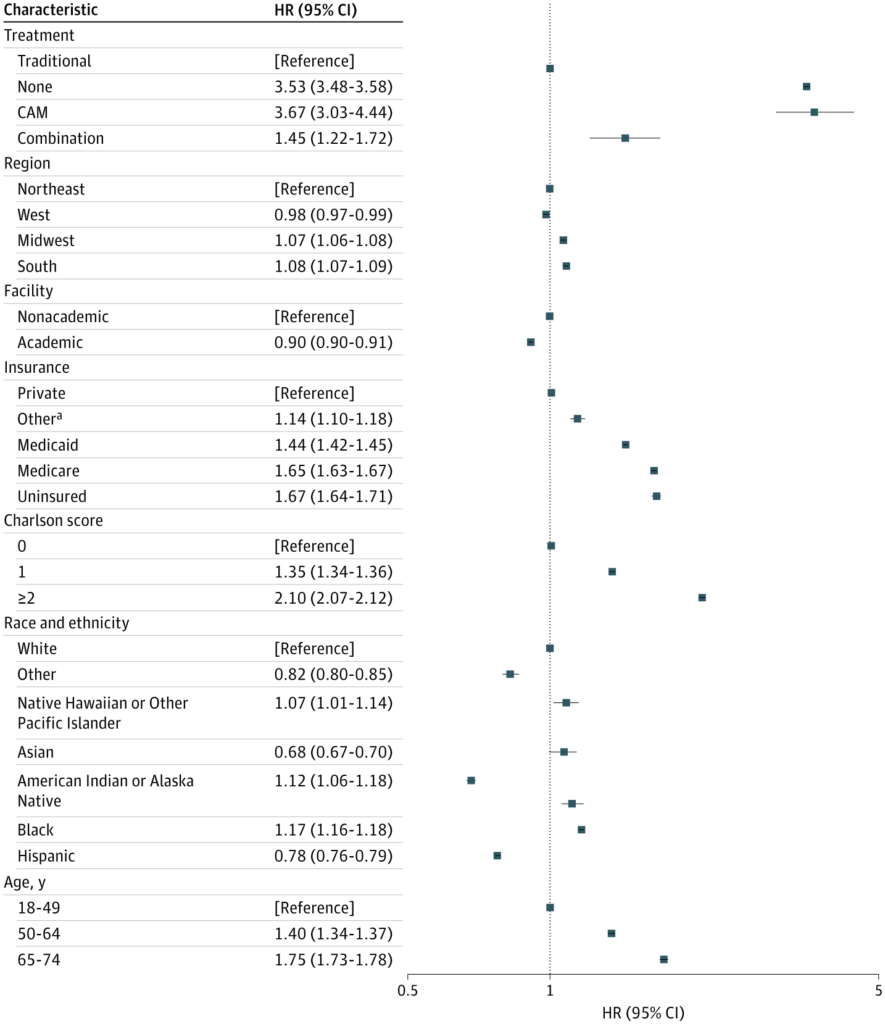
In the adjusted data, both the addition of CAM to treatment and the use of only CAM were associated with an increased hazard ratio for mortality, as was, not unexpectedly, no treatment at all. Interestingly, CAM only was associated with the highest mortality, slightly higher than no treatment at all. Other factors associated with an increased hazard ratio were age, increasing comorbidities, and type of insurance.
The authors also explored why this relationship was observed, specifically the question of whether those who used CAM in addition to SBM skipped one or more modalities among the multimodality treatment of breast cancer, leading to this graph:
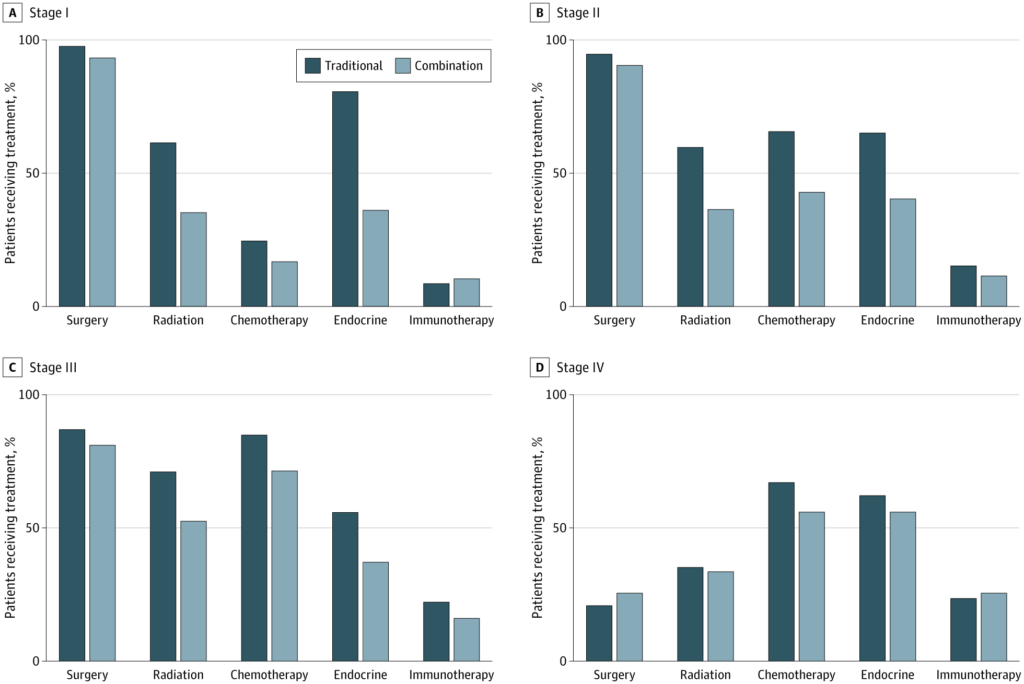
The authors note that surgery was more common among the traditional (SBM) therapy cohort than among the combination cohort, but not by a huge percentage. This tracks with my personal observations, albeit limited, that patients pursuing such “CAM” mostly tend to be accepting of surgery; it’s often the radiation, chemotherapy, and endocrine therapy that they are less accepting of. A more striking difference was observed for radiation therapy, in which those receiving combination therapy underwent radiation therapy less frequently than those receiving standard-of-care SBM treatment for disease that was stage I through III, which are the breast cancer stages for which treatment with curative intent is possible. (Stage IV disease is not curable, only manageable for palliation and delaying death.) Given that, for breast conserving surgery, radiation decreases the risk of recurrence in the breast by as much as five-fold and is also associated with a significant, albeit modest, survival benefit as well.
One thing that puzzled me about this study was that it states:
Receipt of chemotherapy and endocrine therapy was similar between the treatment groups across stages I to III. There were no statistically significant differences in receipt of any particular treatment modality for patients with stage IV breast cancer.
For endocrine therapy, at least, this text sure seems to conflict with what I see in the graph, which appears to show that usage of endocrine therapy was markedly lower among women with stage I through III tumors. (My guess is that the differences that seem to be shown on the graph for usage of chemotherapy are not be statistically significant, given that the sample size of patients who received CAM was only 841 patients.) Endocrine therapy generally consists of strategies that block the action of estrogen for women with estrogen receptor-positive (and therefore estrogen-responsive) tumors, such as tamoxifen, aromatase inhibitors, bilateral oophorectomy, or ovarian suppression. Given that approximately 75% of breast cancers are estrogen receptor-positive and that endocrine therapy can decrease the risk of recurrence by about 40-50% and of death by up to 40% in such patients, this is one possible reason explaining why CAM use could be associated with an increased risk of death. At the very least, there appears to be a large difference in the use of endocrine therapy for stage I patients. (Granted, these are the patients who can be predicted to do the best and have the highest overall survival.) Moreover, I know from experience that endocrine therapy is one of the types of therapies that patients seem to have the most trouble continuing, due to side effects (e.g., hot flashes) over at least five years. (Chemotherapy side effects are definitely worse than side effects from endocrine therapy, but most chemotherapy regimens are less than around four or five months.)
The authors did some sensitivity analyses to try to isolate the effects of omitting one of the recommended “traditional” SBM treatments, using patients with stage II disease:
To better understand the consequences of omitting 1 form of traditional treatment, a sensitivity analysis was conducted as an unadjusted survival analysis for patients with stage II breast cancer, stratified by the receipt of 4 of traditional therapies (chemotherapy, radiation, surgery, and endocrine therapy) (eFigure 3 in Supplement 1). Patients with stage II breast cancer treated with a combination of traditional therapy and CAM had lower survival if they omitted radiation, endocrine therapy, or surgery, but the implications of omitting chemotherapy for survival in this cohort were not statistically significant.
Again, I suspect it’s the small numbers of patients that lead to the impact of chemotherapy not statistically significant in this analysis.
CAM use is still associated with poorer outcomes
So what can we conclude from this study? There has been less research in this area than arguably there should be, but on the other hand doing studies like this is extremely difficult and nearly impossible to do except using databases like the NCDB and retrospective study design. In this case, the authors note:
In this cohort study, we found that the overall survival for patients with breast cancer who received CAM, either alone or in combination with traditional therapy, was shorter than that of patients who received traditional therapy. These findings are consistent with the results of a previous study that found that, when stratified by cancer types, receipt of CAM was associated with worse 5-year survival for breast, lung, and colorectal cancer.24 However, this earlier work had only 123 patients who received CAM for breast cancer and was conducted during an earlier time frame (2004 to 2013). Our study builds on previous findings, has a considerably larger sample size of patients who received CAM (841 patients), and further examines the survival of patients who received a combination of traditional therapies with CAM in subgroup analyses.
Naturally, I discussed that previous study when it was released in 2017. That study also used the NCDB and also used the category in the database of “other-unproven: cancer treatments administered by nonmedical personnel” to identify usage of CAM modalities. Unsurprisingly, it showed worse five year survivals for not just breast cancers but also lung and colon cancer associated with CAM usage. Even though this study examines a much larger sample of breast cancer patients who used CAM, that number is still a very small percentage of the total cohort, leading the authors to speculate:
The relatively low rate of CAM use documented in the NCDB raises a concern that patients may not be discussing their interest in alternative treatments with their oncology teams. Specifically, several studies have estimated considerably higher rates of CAM—closer to 30%—in the breast cancer population.9,44,52 Because the NCDB is limited to capturing CAM use that is documented in the health record, patients may not be including their intentions to use treatment administered by nonmedical personal with their treatment teams. To date, one study found almost half of patients with breast cancer used CAM; however, many of them were not comfortable discussing this with their conventional treatment team because they did not believe that team had enough expertise on CAM.52 Another study found that while many patients choose to use CAM in addition to conventional treatment in the process of their cancer care, clinicians often lack the expertise to counsel patients on the evidence or lack thereof regarding CAM treatments.44 Inviting patients to share their interest in CAM may present an opportunity to enhance shared decision-making, particularly as patients may be planning to forego traditional treatments.
To this I would add that the rise of the pseudospecialty of “integrative oncology,” complete with training programs, seemingly authoritative guidelines endorsed by ASCO, and the proliferation of quackademic medicine in National Cancer Institute-designated comprehensive cancer centers, also means that a lot of patients are getting what would be considered “CAM” treatments that the NCDB wouldn’t capture under a category of “other-unproven: cancer treatments administered by nonmedical personnel,” if only because they’re now being administered by medical personnel at actual cancer centers, under the auspices of ASCO-endorsed SIO guidelines. As is the case with most of these studies, under ascertainment of CAM usage is a problem, although the authors further speculate:
Beyond the traditional limitations that are associated with observational studies, this study has several limitations. The NCDB designation of alternative therapy likely misses many patients who choose CAM, some of which may have experienced meaningfully different outcomes. We suspect that many of these patients may fall within the group with no treatment, which has the worst survival.
May I quibble here a moment? By definition, CAM means “complementary and alternative medicine.” Remember how, back in the day, CAM advocates would argue themselves blue in the face that these modalities were intended to complement, not replace, science-based medicine. That’s a big part of the reason why the term “CAM” eventually gave way to “integrative medicine,” to imply more strongly that these unproven (and in some cases disproven) therapies would never, ever be given except alongside “traditional” science-based medical treatments. (Also, CAM advocates didn’t like including the word “alternative” to describe their favorite woo.) Also, one of the rationales among advocates for embracing “integrative oncology” has been that oncologists and surgical oncologists doing so would make it less likely that patients would forego science-based treatment with large bodies of evidence supporting their efficacy and safety.
I was also less than thrilled with the authors’ apparent No True Scotsman fallacy, namely implying that their study doesn’t show that “true CAM” (i.e., making sure that patients receive every science-based treatment modality that is standard-of-care) impacts survival:
At the same time, because skipping traditional therapies appeared to be common in the combination cohort, a scenario we could not isolate was one in which the patients who fully complied with traditional treatments and then added CAM to the traditional treatment protocol. While the merits of traditional cancer treatments are clear, there are also indicators that treatments that would fall under the category of alternative therapies in the NCDB could have benefits during the cancer journey. Multiple studies have reported on the benefits of CAM approaches for symptom management and quality of life improvement in patients with cancer, including acupuncture,44,45 massage therapy,46,47 and mindfulness-based practices.48,49 Indeed, such therapies are increasingly studied and becoming incorporated into practice guidelines.50,51 Therefore, while traditional therapy should not be replaced by CAM, this study does not present any evidence to support or refute the role of CAM strictly as an adjunct to traditional therapy.
I must point out that, protestations of its advocates otherwise, acupuncture has never been demonstrated to be more than a theatrical placebo, and references cited to support the contention that acupuncture is effective in symptom management consist of nonrandomized self-reported outcomes and the SIO monograph. As for massage, I’ve never really had a problem with massage for making patients feel better; I merely reject the overblown claims that many of its advocates make for it. You can see that what is cited tends to be the commonly cited “CAM” therapies that are probably, as The Hitchhiker’s Guide to the Galaxy famously described the planet earth, “mostly harmless” (which really is the best that can be said about acupuncture) and not a lot of the other “CAM” therapies, such as supplements, herbs, reiki, and a number of other bits of woo and quackery that often fall under the “integrative medicine” label.
All of this brings me back to MAHA. If one looks at how the MAHA movement formed through the coalescence of COVID-19 era antivaxxers, COVID-19 minimizers, and the preexisting alternative medicine movement, it was not surprising in retrospect how rapidly the movement embraced “alternative” treatments for cancer. That’s how a repurposed drug embraced by quacks to treat COVID-19, ivermectin, was soon repurposed again to treat cancer. It’s how so many MAHA-adjacent doctors are sounding more and more like run-of-the-mill cancer quacks from the beforetime. What I fear is that RFK Jr. will build on the pre-existing “integrative medicine” movement that had been growing in influence since at least the mid-1990s, in order to encourage more “integration” of quackery with science-based medicine, not just in oncology but in all medicine. What this study reminds us, once again, is that, just as MAHA vaccine policies will result in the deaths of children, MAHA will likely result in the deaths of cancer patients as well, not to mention just patients.
Crap, I came back around to the devastation that RFK Jr. and MAHA is doing to US public health, biomedical research, and medicine. I guess there’s just no escaping it.

