Various herbal remedies and supplements.
For those of you that missed the Science-Based Medicine day at NECSS last week, I’ve put the highlights in the following post:
The supplement industry is big business, and the popularity of these products seems to keep growing. I once worked at a small independent pharmacy that specialized in supplements, homeopathy and “alternative medicine” as way to differentiate itself from the big chain pharmacies. A lot has changed in a decade. Today, even national chain pharmacies have aisles and aisles of herbal remedies, dietary supplements, and even homeopathy. Yet despite the preponderance of products on store shelves, there is little evidence to suggest that supplements are necessary or even improve health (and may even cause harm). The right for consumers to buy products for themselves, and make their own self-care decisions, is an important one that respects individual autonomy. Regrettably, the regulations of these products are so weak and ineffective that the sale of these products quite possibly harms, rather than helps, the average consumer. The system is rigged against consumers, and it prevents the use of supplements in an evidence-based way.
What exactly is a supplement?
The idea of a risk-free magic bullet that improves our health seems irresistible. Especially when you can choose a product that allows you to avoid what are assumed to be toxic, unnatural, chemical drugs. Supplements are marketed as safe, natural and effective, and there is no question that messaging has been effective. But whether or not a product is considered a drug or supplement may be less clear than you think. But the difference is important, as the distinction can mean:
- Whether there is an evidence to justify their sale
- The presence of any safety studies
- What manufacturers can claim about their products
- The overall quality of the product
Recognizing consumer demand for supplements, and the insistence that these products were self-evidently both safe and effective, many national health/drug regulators have sought to distinguish supplements from “drug products” in some way. The FDA defines supplements as:
A dietary supplement is a product intended for ingestion that contains a “dietary ingredient” intended to add further nutritional value to (supplement) the diet. A “dietary ingredient” may be one, or any combination, of the following substances:
- a vitamin
- a mineral
- an amino acid
- a dietary substance for use by people to supplement the diet by increasing the total dietary intake
- a concentrate, metabolite, constituent, or extract
The FDA’s own definition is problematic. This is a disparate group of products. Some supplements are completely synthetically manufactured, such as certain vitamins. These products are probably the closest to drug products (and in some cases, are considered drugs). They can have specific and genuine, established uses (e.g., folic acid in prenatal vitamins for preventing neural tube defects). These products can be easily manufactured to the same standard as conventional drug products. But the category of “supplement” also includes herbal remedies – which I would argue shouldn’t be “supplements” at all. There is no routine need to supplement the diet with any “herb or other botanical”. Outside of cooking, these products are consumed for what are believed to be medicinal effects. Yet an herbal remedy has very different regulatory oversight than if we purified the same herb, isolated the active ingredient, and manufactured it. Then it would be a drug, not an herb. Herbal remedies are effectively crude, unpurified drugs – possibly with hundreds of ingredients, and unclear product standards. Many drugs sold today have botanical origins, but are not considered supplements. Acetylsalicylic acid (aspirin) is derived from salicin, found in the bark of the willow tree. Atropine was found in nightshade, and digitalis (digoxin) is what makes the foxglove plant toxic. Yet they’re drugs, not supplement.
If the US approach to regulation sounds artificial, you’re right. For more information on the Dietary Supplement Health and Education Act of 1994 (DSHEA) that regulates supplements, there are multiple other posts that explain it in more detail.
When is a supplement not a supplement?
In general, supplements are considered safe until proven unsafe. Regulation is light, as mandated by DSHEA. In contrast, drugs are strictly regulated and must be shown to be both safe and effective before they are allowed for sale. But is the line really that clear? Consider these two examples.
- Vitamin A is sold as both a supplement and a prescription drug, depending on the formulation and the dose. A routine ingredient in multivitamins, it’s also used as a drug to treat acne, cancer and anemia. Vitamin A is not innocuous, either. Side effects can include birth defects and liver toxicity.
- Melatonin is a naturally-produced hormone that’s also found in some foods. Supplements are typically manufactured synthetically (and are safer in that form) and are the only hormone available without a prescription in the United States. There’s some evidence for effectiveness with jet lag, but it’s used for a variety of sleep/wake disorders. The long-term effects, particularly in children, are not well understood.
Drug or supplement, there are some common goals
Regardless of whether you’re taking a drug or a supplement, you typically want the following to occur:
- The product to be absorbed in the body
- That substance to get to where it needs to in the body
- That substance to have some sort of a beneficial or positive effect
- That substance to not be harmful to us, meaning minimal side-effects or other toxicity
In order for any of this to occur, we need to consider the science of pharmacokinetics. It’s not necessary to understand the intricacies, but a general understanding of the four factors that influence how a substance behaves in the body when we ingest it can help us evaluate the plausibility of efficacy claims. The shorthand is ADME, for absorption, distribution, metabolism and elimination (more here, for you pharmacokinetics nerds):
- Absorption: What you swallow does not automatically equal what your body will absorb. Whether or not a drug or supplement is absorbed depends on the route of administration, the formulation (capsule/tablet/liquid/raw material), the chemical properties of the substance, and other factors (like food in the stomach). It needs to survive being dispersed in stomach acids and digestive enzymes. It must then dissolve fully into gastric juices (particulate matter is not absorbed by the body), and then the substance must cross cell membranes – just to enter bloodstream. Not all substances are absorbed. Drugs like insulin must be injected because they are destroyed in the stomach. Even if it makes it as far as absorption, a substance then needs to survive the “first pass effect“, which is passage of blood through the liver before it reaches the rest of the body. In the liver, enzymes attempt to eliminate and metabolize molecules, deactivating anything foreign or unwanted.
- Distribution: Once a substance is absorbed, it then has to be distributed in the body to the location where it will have its presumed effect. You cannot simply assume a substance will reach any part of the body to any significant degree. A substance may adhere to proteins in the blood, reducing the “free” amount that is available for distribution to other cells. Not all chemicals, (drugs and supplements alike) are distributed equally to all parts of the body – some may deposit more readily in fat tissue, for example. The blood-brain barrier blocks most foreign molecules from reaching the brain.
- Metabolism: Foreign or waste substances are typically excreted by the kidneys in the urine, but can also be secreted in the bile. Molecules may undergo chemical changes, facilitated by enzymes in the liver (and elsewhere) to make them easier for the body to eliminate. This transformation or “metabolism” is true detoxification (not the fake detox). As your ability to metabolize a substance can vary based on factors like age and genetics, drugs (and supplements) can also have their effects modified. Importantly, drugs and supplements can be metabolized by common pathways, which can lead to a drug-supplement “interaction” where the action of one substance is modified by another.
- Elimination/Excretion: Foreign substances need to be eliminated from the body, otherwise blood concentrations could potentially rise to dangerous levels. The kidneys are the main organs of excretion, but secondary roots can include the stool, lungs, skin, and even breast milk.
The net effect can be illustrated in the classic concentration/time graph: Note the gradual onset (due to absorption), the peak level in the bloodstream, and then the effects of metabolism and elimination, as the concentration drops off again:
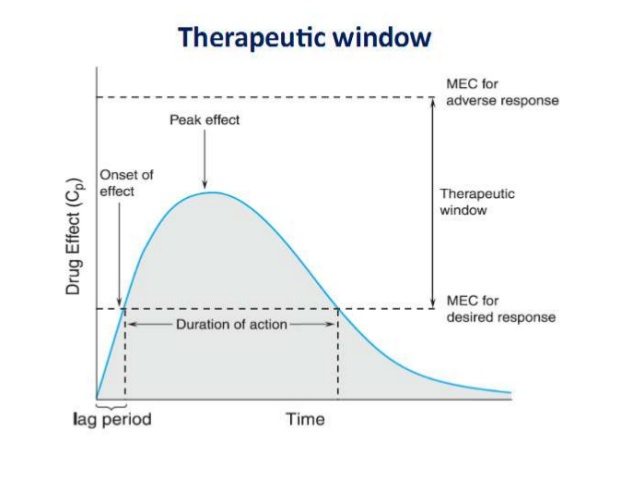
The same kinetics rules apply
The concentration-time graph is equally applicable to drugs and supplements. Consider vitamin D, which is considered to have a wide “therapeutic window”, meaning it’s nearly impossible to overdose on the vitamin to the point where you’d have side effects. Supplement seller Gary Null found a way, however, when he was poisoned by his own brand of supplements which was later found to contain excessive amounts of vitamin D.
Another example I like to cite is oil of oregano. Reading the marketing, you might think this oil has magical healing properties, from toenail fungus to skin infections, to what are claimed to be simultaneous antiviral, antibacterial and antiparisitic effects. And there are in vitro (test tube) studies showing that this product may have some medicinal properties. But in order for oil of oregano to have an effect in the body, it must be absorbed and distributed to the site of action where it must be present in sufficient quantity (and for an adequate duration) to have an effect. As a corollary, consider how you clean your hands: Alcohol hand gel is also antiviral and antibacterial, but no matter how many maragaritas you may drink, you will never be able to raise your blood alcohol level high enough to kill bacteria or viruses- without killing yourself first. The dose matters. So considering the claims about oil of oregano, claims such as “disinfectant” or even “skin infection treatment” are far more plausible than claims it’s effective for internal infections. There’s simply no evidence to suggest that the chemical constituents of oil of oregano are absorbed and available in the body to have any effect at all.
Drugs can have interactions with each other, and drug-supplement interactions are also common. Supplements can affect the ADME of drugs, leading to potentially harmful effects:
- Calcium supplements can bind to drugs in the gastrointestinal tract and reduce overall absorption.
- Quercitin can displace the protein binding of “blood thinners” like warfarin, potentially increasing bleeding risk.
- Ginseng is among many supplements that can modify the way liver enzymes work, interfering with the way drugs work.
There’s also the risk of additive effects, where a drug and a supplement may have similar effects: concurrent use of vitamin E, ginkgo biloba, warfarin and aspirin can increase the risk of bleeding over drug therapy alone.
Supplements have unique risks
Now that we’ve established the common chemical and biological properties that apply to drugs and supplements, it’s worthwhile noting that there can be important differences in their characteristics as well. One of the most important limitations of supplements, in particular with the herbal and botanical-based remedies, is that there may not be any identified active ingredients. This may not initially sound concerning, but it has dramatic implications on how we assess efficacy, and the relevance of different clinical trials. If there has been no basic research that’s identified the one (or more) active chemicals among the hundreds of chemical that may be in a herb or plant supplement, it is simply impossible to apply basic quality control standards. Are you getting a leaf, a stem, the flower, or the root? When was it harvested? How was the product prepared? Without a known active ingredient, measuring blood concentrations can’t be done – because you won’t’ know what to measure. Consequently, herbs and botanicals could potentially vary in their chemical makeup from brand to brand and even batch to batch of the same product.
Contrast the herbal remedy with the typical drug product, such as a new chemical in development. Because drugs must be tested before they are allowed for sale, the chemical characteristics (ADME) must be known and well understood before any tests of efficacy. The quantity per tablet or capsule is measured precisely, and can be verified. When any clinical trial is conducted, the same formulation (e.g., capsule or tablet) is used. If that drug is among the molecules that eventually makes its way to a pharmacy near you, the manufacturer must demonstrate that the version that is sold behaves identically in the body as the formulation that was tested in the clinical trial. This gives your physician, your pharmacist, and you the confidence that the clinical trials that established a drug’s efficacy are relevant to the product you have been prescribed. None of this is possible with herbal remedies. Quality manufacturing standards are rare, and standardization is uncommon. Is a clinical trial with a supplement relevant to the product you’re considering buying? There’s simply no way to know.
Safety concerns are significant as well. Few supplements are actually subjected to rigorous clinical trials where safety can be measured. Harms are rarely reported to regulators – if they are even detected. The story of Aristolochia is cautionary and telling. Aristolochia is a plant that has been considered to have medicinal properties for hundreds, perhaps thousands of years. Owing to its resemblance to a uterus it has been thought to be effective for childbirth and has been used as part of traditional Chinese medicine for a number of conditions, (none of which were supported by any evidence). When a cluster of Belgian women developed kidney disease and urinary cancers after taking Aristolochia-containing diet pills, the catastrophic harms from this product were revealed. Aristolochia promotes cancer formation through DNA mutations. It’s now classified as a type 1 carcinogen, alongside asbestos and solar radiation. Are other botanicals or supplements causing other harms? There’s no routine data collection, so it’s impossible to say for sure. And even if there was, drawing associations is exceptionally difficult: The appearance of an effect like cancer may lag for years, or decades. The harms of a product like Aristolochia are so widespread and diffuse that it’s notable that it took a cluster of cases in Belgium to finally bring this toxicity to light. We may see elevated number of urinary cancer cases in some Asian countries, possibly for decades. Aristolochia is an example of the challenge we face in identifying and monitoring for adverse effects from herbal products. We simply have no clear understanding of the potential for harms, which is magnified when we can’t even be confident that what’s on the label is actually in the bottle.
Identity, purity, potency and stability questions
To ensure product consistency, we need to be assured that the main ingredient has been tested and is in fact present in a consistent dose. We need the assurance that there are no contaminants present. We need to be sure that the potency hasn’t decreased over time due to the preparation of the dosage form. Finally, we need to be sure that the supplement is stable when it’s stored. It’s notable that the FDA doesn’t mandate expiry date studies with supplements – because with herbals and botanicals, it’s not even clear how you’d establish a product is no longer potent. This is much easier with multivitamins and minerals.
Is your supplement manufactured to high quality standards?
While supplement manufacturers are expected to meet the FDA’s Good Manufacturing Practices (GMP) standards, there’s little evidence that supplement manufacturers consistently do this. FDA inspections repeatedly show that few companies are doing everything they can to ensure products are of high quality. Consider the following statements, with emphasis added.
From 2012, Trine Tsouderos noted in The Chicago Tribune:
Federal inspections of companies that make dietary supplements — from multivitamins and calcium chews to capsules of echinacea and bodybuilding powders — reveal serious and widespread manufacturing problems in a $28 billion industry that sells products consumed by half of all Americans. In the last four years, the U.S. Food and Drug Administration has found violations of manufacturing rules in half of the nearly 450 dietary supplement firms it has inspected, according to agency officials. The inspection reports portray an industry struggling to meet basic manufacturing standards, from verifying the identity of the ingredients that go into its products to inspecting finished batches of supplements.
From May 2013, from Naturalproductsinsider.com, an industry publication:
During a three-year period, federal inspectors have cited seven out of 10 facilities that make dietary supplements for violating regulations intended to ensure products are safe and contain the ingredients that are listed on the labels.
And:
“Unlike traditional foods, when it’s rotten, the consumer will often know by taste or smell, [but] with supplements we’re swallowing them based on blind faith in the manufacturer,” noted Pieter Cohen, M.D., a general internist at Cambridge Health Alliance and assistant professor of medicine at Harvard Medical School, who has studied supplement safety and reviewed the FDA data. “But FDA inspections appear to reveal a different story: when we take a look behind closed doors, less than a third of companies are doing everything they can to ensure top-notch products.”
From March 2016, also from Naturalproductsinsider.com, the problems don’t appear to be resolved, based on FDA inspections:
The reinspection data still reflects continuing areas of concern for the industry. Fifteen dietary supplement reinspections—or nearly one-quarter of all of them—resulted in citations for failure to establish product specifications. Setting product specs, cGMP consultants have noted, is a basic requirement. Around 17 percent of reinspected firms were cited for having inadequate batch production records.
“Largely, it’s the same problem … that systematic issue with GMP compliance,” FDA’s Welch said, citing the most common violations in the reinspection data.
“As we’ve seen, the industry knows we need to move much further beyond cGMPs to have a product that consumers can trust,” said Pieter Cohen, M.D., an assistant professor of medicine at Harvard Medical School who has researched a number of sports products that are tainted with illegal substances. “So the fact that 60 percent of firms inspected aren’t even making this incredibly low bar of cGMP is frankly embarrassing and poses serious risks to consumers.”
And also:
Joy Joseph, a cGMP consultant to dietary supplement firms, said many new companies have emerged in the dietary supplement industry. She still encounters firms that aren’t even aware of the FDA regulations in spite of the fact that they were adopted nine years ago.
“We still have a lot of companies that have never been inspected, and still today, most dietary supplement companies don’t have laboratories, and they don’t have qualified people to establish the correct specifications and do the testing,” said Joseph, principal consultant with Joy’s Quality Management Systems, in a phone interview.
“And at the end of the day,” she added later, “it is the consumer who is being harmed by these products being manufactured by companies who don’t know anything about GMPs.”
What we’re left with, as health professionals and consumers, is an industry without the ability to manufacture products that meet the basic standards in place for pharmaceuticals. Moreover, the evidence suggests that some of these companies are not even aware of the expectations to make products that meet basic standards for identity, purity, potency, and stability.
Adulteration is not uncommon
I and others have blogged multiple times about what appears to be the routine adulteration of supplements. Vox has a great website where you can check if your supplement may have drug contaminants:
These pills are barely regulated. Supplement makers don’t need to prove their products are safe or even effective before putting them on store shelves. And while supplements are supposed to be accurately labeled, a Vox review of government databases, court documents, and scientific studies uncovered more than 850 products that contained illegal and/or hidden ingredients — including banned drugs, pharmaceuticals like antidepressants, and other synthetic chemicals that have never been tested on humans.
We found examples of weight loss supplements spiked with cancer-causing drugs that had been pulled from the US market, and brain enhancers laced with chemicals that have never been approved for sale in the US. More than 100 products contained DMAA, a drug that’s been banned in the US, UK, and several other countries because it is linked to strokes, heart failure, and sudden death.
Several months ago I described an analysis of 26 traditional Chinese medicine products that found 92% were contaminated with pharmaceuticals, heavy metals, or the DNA of animals, including traces of cat, dog, rat, or pit viper. I’ve also described actions of the New York State Attorney General’s office. It accused four large retailers of selling supplements that failed to contain labelled ingredients. Testing the products with a technology called “DNA barcoding”, the AG’s office concluded that most of the products contained little to none of the labelled ingredient. They also found ingredients that were not disclosed on the label. While this testing technology was criticized (fairly) for its technical accuracy, it’s notable that vitamin retailing giant GNC has agreed to implement its own DNA barcoding testing technology for its products, and will be setting a higher quality standard that what is required of the industry.
How can we select and use supplements safely?
A supplement worth taking is backed by good evidence, and is unlikely to cause harm. Most supplements fail this test – usually because they lack good evidence to show they work. But even where the evidence is promising (and the risks appear acceptable), there’s an additional consideration with dietary supplements – supplement quality is unclear. When you consider the different products that are considered supplements, multivitamins and minerals are probably the safest product – the ingredients are known, they can be measured, and the manufacturing can be standardized. There’s more risk with the herbal and botanical remedies. Not only is there a lack of consistency, there’s a much greater risk of adulteration and contamination. Finally, sexual enhancement, weight loss, and body building supplements seem most likely to be adulterated (and there’s no reason at all to take these products). When it comes to dietary supplements, there’s no easy way for a health professional or a consumer to independently verify that a product is of high quality. Regulatory structures leave a lot to be desired, resulting in a “buyer beware” marketplace for consumers. Until something changes, it will continue to be nearly impossible to have confidence in the quality of supplements, or to use them in science-based ways.