The dietary supplement market in the United States is a multi-billion-dollar industry, enjoying steady growth year over year, and is currently valued at over $40 billion. The industry encompasses a wide range of products, including vitamins, minerals, herbal remedies, sports nutrition supplements, and more. Fish oil supplement sales are big business, and the category is one of the more popular in the market. Annual sales of fish-derived supplements amount to more than a $1-billion market in the United States, driven more by hype than good evidence. Many formulations are sold over-the-counter, while others require a prescription or are available for purchase from a health care provider. The enthusiasm for fish oil has been influenced in part by observational studies of diets, noting a relationship between fatty fish consumption and cardiovascular disease. But in trials with fish oil capsules, those benefits haven’t been demonstrated.
The absence of good product quality and labelling standards is a substantial barrier to the science-based use of supplements. With drugs, standards are rigorous and largely consistent across countries: Clinical trials are conducted to determine if a drug has meaningful effects. These clinical trials are scrutinized by regulators. Products approved are considered safe and effective based on that evaluation. Marketing claims related to drugs are permitted only as they are consistent with the evidence that supported their approval.
The same quality assurance standards are not in place for supplements. Even if there is promising data from clinical trials, we can’t make the same inference about the expected effects, because there is not guarantee that a supplement for sale is identical to (or even closely similar to) another product.
A new paper by Joanna N. Assadourian and colleagues looks at health claims and doses of fish oil supplements in the US. Published in JAMA Cardiology in August 2023, it reinforces existing concerns about the labelling of supplements, and gives further evidence that consumers are not well served by the status quo.
Fishy claims
The US Food and Drug Administration (FDA) is responsible for overseeing the regulation of health-related statements found on supplement labels, as mandated by the Dietary Supplement Health and Education Act of 1994. For fish oil supplements, two categories of health claims are permitted: qualified health claims (QHCs) and structure/function claims (SFCs).
QHCs are statements that pertain to the potential of a supplement or food product to treat or prevent diseases. QHCs are vetted by the FDA against the evidence. (It’s worth noting that all QHCs include qualifying language that acknowledges the absence of a universally established scientific consensus or the presence of uncertainty.) In the case of fish oil, there are two FDA-approved QHCs, one addressing coronary heart disease (CHD) and the other focusing on blood pressure (BP), both in 2004. However, since that time, RCTs have failed to show that fish oil has cardiovascular benefits. The FDA does continue to allow these claims, despite the accumulated evidence since 2004.
SFCs claims describe the role of a nutrient or dietary component in influencing the structure or function of the human body. Importantly ,these claims cannot state that the supplement can prevent, treat, or cure any disease. Examples of structure/function claims include statements like “calcium contributes to strong bone health” or “fiber supports regular bowel function.” If you see phrases such as “maintains,” “supports,” or “promotes” on a supplement label, that is an SFC.
Perhaps given there is no obvious or proven benefit to fish oil supplements, products are sold in a variety of formats and doses. When administered at higher levels, eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA), the active constituents found in fish oil, are plausibly beneficial. In an RCT, a daily intake of 4 grams of purified EPA led to a reduction in cardiovascular events. However, a subsequent trial involving high-dose combined EPA and DHA did not demonstrate any cardiovascular benefits. When consumed at doses of 2 grams or more per day, EPA and DHA can lower triglyceride levels in individuals with hypertriglyceridemia, albeit at an elevated risk of atrial fibrillation.
The authors in this study sought to evaluate the claims made on labels of fish oil supplements and to examine the doses of marketed supplements. It used labels from fish oils taken from the National Institutes of Health Office of Dietary Supplement’s Label Database. This database is compiled of product labels voluntarily submitted by manufacturers. Manual data extraction was used to determine the total daily dose of EPA, DHA, and total EPA+DHA from each supplement. Screening of each label was performed to to find and categorize QHCs and SFCs.
Starting with an initial list of of 20,000 supplements in the database, the final data set included 2819 fish oil supplements, of which 2082 (73.9%) contained at least one health-related label claim. The overwhelming majority of claims where SFCs (80.8%), 18.9% made a QHC for coronary heart disease and 0.14% used a QHC for blood pressure. The majority of labels suggest advantages for cardiovascular health, such as statements like “supports heart health.” Many fish oil supplements also assert benefits for various other bodily systems, such as brain/mental health, joint health, and eye health, despite a lack evidence to substantiate these claims. Claims were distributed as follows:
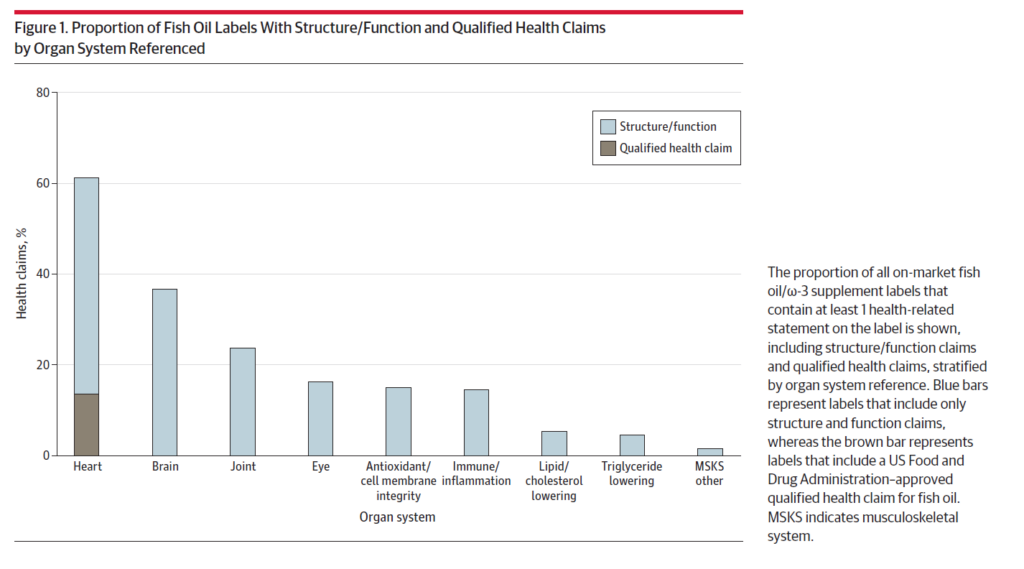
Some of the SFAs on labels included:
- Heart: “promotes heart health”; “supports heard, mind and mood”
- Brain: “DHA is one of the good fats that helps keep our brains running optimally as we age”; “helps prevent against normal cognitive decline as we age”
- Joint: “promotes joint comfort & mobility”; “omega-3 fatty acids are necessary for many vital functions in the body and support heart health and healthy skin and joint function”
- Eye: “maintains eye health and normal vision”; “promotes brain, vision, joint and heart health”
- Antioxidant: “powerful antioxidant support for healthy aging and vitality”; “broad spectrum cellular health formula”
- Immune/inflammation: “immune system support”; “advanced cellular support & immune activation”
- Lipid lowering/cholesterol lowering: “Omega 3 FA, especially when combined with exercise, can help to support the maintenance of healthy blood lipids”
- Triglycerides: “clinically proven to maintain healthy triglyceride levels”
Looking at dosing in a subset of products, 255 labels were examined. Most (91.2%) contained EPA and DHA, with a minority containing one or the other. The median dose was 240mg of EPA and 270mg of DHA, and a total of 600mg/day of combined DHA and EPA. Few (9.4%) had doses of 2000mg or more of EPA+DHA which was the higher dose necessary to lower triglycerides.
Navigating the supplement oil: Buyer beware
There were several limitations to this study. Importantly, it was based on labels that were voluntarily submitted to the National Institutes of Health Dietary Supplement Label Database. There could be other products where the manufacturer didn’t bother submitting a label – and those were not captured. For dosages measurements, only supplements from the 16 largest potential brands were evaluated.
The primary and most important finding of this audit of fish oil product labels was that they showed a diversity of structure/function claims, which are not supported by robust evidence. While it’s not known if unfounded claims are driving demand for this category of supplements, consumers are likely not well-served by products with a variety of unsubstantiated claims. This study also showed a array of product formulations and doses. Unfortunately there was no actual audit of capsule content, so it’s not known how accurate labels actually are. While the authors point to this variety of product formulations (and doses) as a problem, I personally feel that as long as products are accurately labelled, then a diversity of formulations is reasonable. However, it needs to accompanied by good information for consumers so they know what an “appropriate dose” is. Given there is a lack of convincing evidence demonstrating that that fish oil capsules are beneficial for health, perhaps this is an unrealistic expectation.