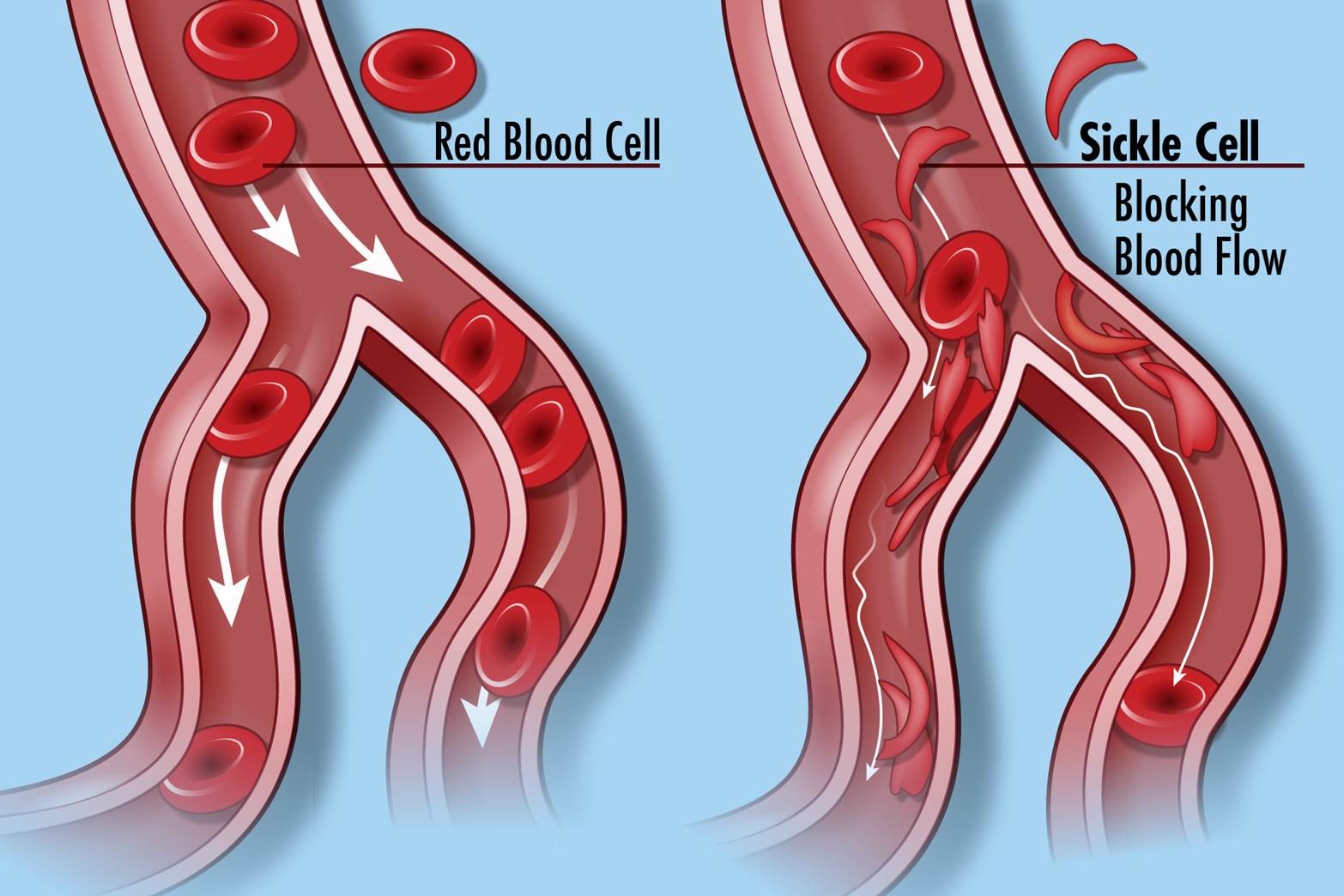
Sickle cell disease is a serious genetic disorder, an abnormality of hemoglobin that is inherited in an autosomal recessive pattern, where both parents have to pass on the defective gene. The red blood cells take on a sickle shape and obstruct small blood vessels. In areas where malaria is prevalent, sickle cell hemoglobin can improve survival from malaria, but where malaria is absent it only causes harm. Patients develop painful crises that often require inpatient management and opioids; they develop acute chest syndrome with pulmonary infiltrates; and they may develop avascular necrosis with the death of bone. They are anemic, with low levels of hemoglobin. They can develop complications including bacterial infections, strokes, gallstones, spleen malfunction, and even infarction of the penis. Life expectancy is reduced: 42 years for males and 48 years for females.
Current treatments include hydroxyurea, blood transfusions, bone marrow transplants, and allogeneic stem cell transplants from a matched sibling donor. The disease can be cured if the patient can switch to producing normal hemoglobin in place of the defective form. Research is ongoing, with trials of umbilical cord blood transplants and gene therapy.
While awaiting a cure, Big Pharma has stepped up to the plate with new drugs, but it has struck out. It gained FDA approval to market two new drugs, Crizanlizumab (Adakveo) and Voxelotor (Oxbryta). The Medical Letter recently reviewed these two drugs, and the news was not good. Crizanlizumab is a monoclonal antibody that binds to a protein expressed on the surface of endothelium, inhibiting adhesion, increasing microvascular blood flow, and reducing vaso-occlusion. Voxelotor inhibits the polymerization of sickle hemoglobin.
The evidence from clinical studies
Crizanlizumab was tested in 198 patients in a double-blind trial of patients who had 2-10 sickle cell related pain crises in the previous year. The annual rate of pain crises was 1.63 with crizanlizumab and 2.98 with the placebo. Statistically significant, but not very impressive. It is given by IV infusion over 30 minutes every four weeks, and a year’s treatment costs $122,571.30.
Voxelotor was evaluated in the double-blind HOPE trial of 274 patients who had 1-10 vaso-occlusive crises in the previous year. The primary endpoint was an increase in hemoglobin of >1.0 g/dl at week 24. 33% responded to the 900 mg dose, and 51% responded to the 1500 mg dose, compared to 7% with the placebo. The Medical Letter pointed out that “Whether voxelotor decreases the frequency of sickle cell crises and incidence of multiorgan failure and death remains to be determined.” Voxelotor tablets are taken orally once daily. The cost for a year’s treatment is $126,739.70. Numerous adverse effects were reported with both of these drugs.
Were you underwhelmed by the evidence and overwhelmed by the cost? I was. So was The Medical Letter. Their conclusion:
In clinical trials in patients with sickle cell disease, crizanlizumab-tmca (Adakveo), which prevents vaso-occlusion, reduced the number of painful vaso-occlusive crises and voxelotor (Oxbryta), which inhibits polymerization of sickle hemoglobin, increased hemoglobin levels, but clinical data are limited and both drugs are prohibitively expensive.
High drug prices are a problem
This is another in a long list of new drugs that offer small benefits at large prices. Is it worthwhile to pay for them? How much is one fewer sickle cell crisis a year worth? Are the drug companies actually going to make a large profit on them? They are not covered by Medicare and have only temporary approval from some insurance companies. Science can give us information about the efficacy of these drugs, but difficult decisions will have to be made by society as a whole and by individual patients whose willingness to take risks may not match their financial resources. There will be cost-benefit analyses, and factors like poverty and racial bias will have to be considered. And prices are high not only for new drugs, but for older, life-saving drugs like insulin and epi-pens whose price has risen beyond many patients’ ability to pay. I don’t have any solutions, but I have a lot of concerns.

