Well, the latest round of grant applications and pre-applications is finally over, which gave me time this weekend to peruse the stack of journals that’s been accumulating on my desk. Oddly enough, despite my being about as plugged in as you can be at my age, I’m still old-fashioned enough to enjoy the physical sensation and the overall experience of picking up the most recent issue of a journal and randomly flipping through it. There’s something about the feel of the paper, the smell of the coating and print, as well as the sheer undirectedness of it all. It’s how I find articles that I probably would never find if I relied just on perusing the table of contents of an electronic edition.
Sadly, that’s not how I found this week’s topic. The study that I’m going to discuss this week is an E-pub ahead of print that I became aware of through a Reuters story, late last week when I still didn’t have time to deal with it. As is my usual practice, I saved the link for later in Safari’s Reading List, and this time I actually managed to come back to it. The story is entitled “Many cancer patients expect palliative care to cure“, and it’s about a recent publication out of the Dana-Farber Cancer Institute published online in the Journal of Clinical Oncology (JCO) entitled “Expectations About the Effectiveness of Radiation Therapy Among Patients With Incurable Lung Cancer“. It caught my eye, even a week ago, because managing expectations in patients with advanced cancer. It then led me to do a search for related articles, which brought me to a similar study from last fall in the New England Journal of Medicine, entitled “Patients’ Expectations about Effects of Chemotherapy for Advanced Cancer“, also from the same group at the Dana-Farber. This latter study looked at patients’ expectations regarding chemotherapy, and I even remember having encountered it when it was first published and wanted to blog about it then. I don’t recall why I didn’t, but here’s my chance to revisit it.
There is not a cancer clinician out there whom I’ve ever met who hasn’t thought that he’s given a patient with incurable cancer an accurate, science-based assessment of his prognosis, only to find that the patient still expects to be cured. Even as a breast surgeon, I occasionally encounter the issue even though the vast majority of my patients are early stage and therefore potentially curable by surgery, supplemented with a combination consisting of one or more of the common breast cancer treatment modalities of chemotherapy, hormonal therapy, and radiation. In my practice, it’s usually when I’m referred a patient who has stage IV disease that has either been stable for a while, leading to the question of whether removing the primary tumor will prolong life. There is evidence that it will, but, given that the data supporting this contention are all largely retrospective and no large randomized trials have been published yet, many of us suspect that there is a large component of selection bias in the existing data supporting removing the primary tumor in patients with metastatic disease.
The other example is the occasional patient with metastatic disease whose primary tumor is large and on the verge of ulcerating. Disease that gets to this point is known as en cuirasse disease. It’s nasty in the extreme, leaving a woman with a bleeding, stinking, necrotic mass on her chest wall that can’t be removed without major surgery that involves removing huge swaths of skin, soft tissue, and chest wall. Such surgery is rarely, if ever, done, because it’s too major to be proposed for palliation of a patient who likely only has months to live. If you want an example of just how horrible a death comes from en cuirasse disease, I described one at the end of my post on German New Medicine, specifically the example of Michaela Jakubczyk-Eckert. (Warning: That link is not for the squeamish.) Surgery is often worth it to prevent such a complication of advanced breast cancer, even if it won’t save the patient’s life. I try to be very clear—blunt, even—about this whenever I see the occasional patient referred for consideration of palliative mastectomy, but, even so, although I can’t give a percentage a significant number of patients appear to believe that having a mastectomy might actually save their lives.
So it is with patients with metastatic lung cancer and colorectal cancer receiving chemotherapy, as illustrated in the NEJM article. In both studies, which are based on the same national, prospective, observational cohort study, the Cancer Care Outcomes Research and Surveillance (CanCORS) study, a large multi-center consortium study funded by the National Cancer Institute whose aims are to:
- assess the quality of care received by cancer patients and survivors;
- explore the relationship between quality of care and patient health outcomes; and
- evaluate how characteristics of patients, providers, caregivers, and delivery systems affect quality of care and outcomes.
Specific questions to be answered include:
- Are there racial, ethnic, and socioeconomic differences in use of effective therapies for colorectal cancer and lung cancer?
- Are there racial, ethnic, and socioeconomic differences in patients’ assessments of the quality of cancer care?
- How do patients and physicians decide about therapies for colorectal cancer and lung cancer?
- Why do outcomes of lung cancer surgery and colorectal cancer surgery vary by hospital and surgeon volume?
- What factors explain participation in clinical trials for lung cancer and colorectal cancer?
- Are patients’ symptoms recognized and treated effectively at different stages of illness?
Obviously, these two papers report on the results of asking about how patients and physicians decide on therapy for colorectal cancer and lung cancer and racial differences in patients’ assessments of the quality of their cancer care. Specifically, the part of the study that I’m going to discuss involved assessing patients with newly diagnosed metastatic lung or colorectal cancer (stage IV) who were alive four months after diagnosis and had received chemotherapy (the NEJM study) or who had newly diagnosed lung cancer, stage IV or stage IIIB with malignant pleural effusion, who had received, or were scheduled to receive, palliative radiation therapy. There were 1,193 patients in the NEJM study and 384 in the JCO study, both respectable-sized cohorts. The questions asked were adapted from the Los Angeles Women’s health study and were worded:
After talking with your doctors about chemotherapy, how likely did you think it was that chemotherapy (in the case of the NEJM study) or radiation therapy (in the case of the JCO study) would:
- help you live longer?
- cure your cancer?
- help you with problems you were having because of your cancer?
Response options were “very likely,” “somewhat likely,” “a little likely,” “not at all likely,” and “don’t know.” Refusal to respond was also recorded. Patient demographics were, of course, recorded and correlated with the likelihood of various responses.
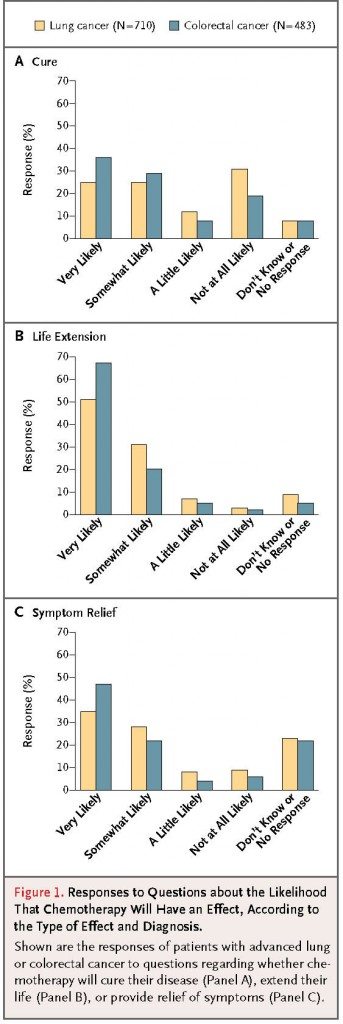
In the part of the study reported in the NEJM, I think it’s easiest just to present the “money graph.” There are three panels: A. Cure; B. Life extension; and C. Symptom relief. The results are:
The authors described the results thusly:
The proportion of patients with inaccurate expectations about the likelihood that chemotherapy might cure their cancer according to patients’ characteristics is shown in Table 1. Overall, 69% of patients with lung cancer and 81% of those with colorectal cancer gave answers that were not consistent with understanding that chemotherapy was very unlikely to cure their cancer.
This is a huge number, and the authors reported that the likelihood of misunderstanding correlated with a diagnosis of colorectal cancer as opposed to lung cancer, nonwhite patients or ethnic group (specifically Latino and Hispanic patients) as compared to white race (odds ratio, 2.82, confidence interval 1.51 to 5.27), African-American race (odds ratio 2.93), and Asian or Pacific Islander patients (odds ratio, an amazing 4.32). No other factors, including education, functional status, and the patient’s role in decision making, correlated with a higher likelihood of an inaccurate response about the curative potential of chemotherapy.
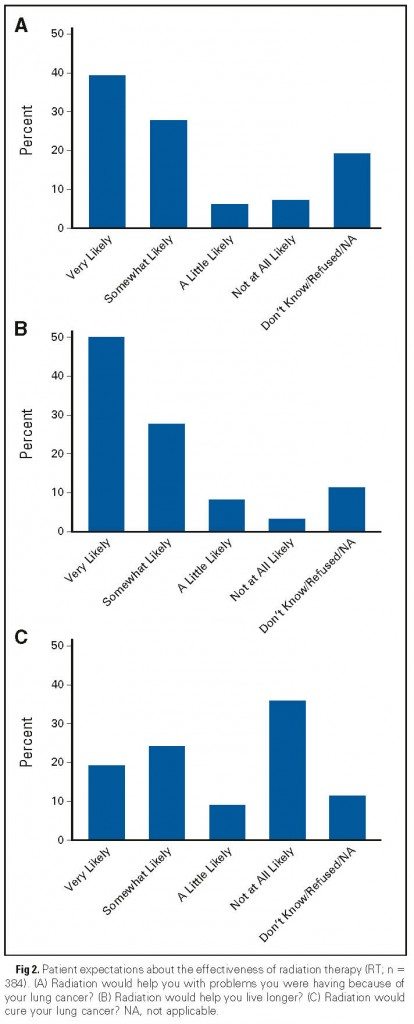
But what about radiation therapy? The JCO study reported depressingly similar results. Here is the equivalent graph to the one above for patient understanding about the curative potential of radiation therapy. Note that, for reasons unclear to me, the order of the questions are reversed: A. Symptom relief; B Life extension; and C. Cure. I don’t know why. The authors just chose to do it that way.
Depressingly as well, the authors’ conclusion about radiation therapy is very similar to their conclusion about chemotherapy, except that slightly different factors appear to be correlated with an inaccurate understanding of the palliative nature of the proposed treatment:
The proportion of patients who did not accurately understand that palliative RT was “not at all likely” to cure their cancer was 64%. Table 2 shows the unadjusted and adjusted analyses of factors associated with inaccurate beliefs about cure from RT. On multivariable analysis, older patients were more likely to have inaccurate beliefs (with age younger than 55 years as the reference, odds ratio [OR] was 1.44 for ages 55 to 64 years, 1.78 for ages 65 to 74 years, and 2.45 for ages older than 74 years; overall P =.02) as were patients of nonwhite race (African American OR, 1.48; other nonwhites OR, 3.32; overall P =.009), and patients whose surveys were completed by surrogates because they were too sick were less likely to have inaccurate beliefs (OR, 0.54; P = .04). Likelihood of inaccurate beliefs also varied by PDCR site (overall P .04).
Then, combining the results about radiation therapy with the results concerning chemotherapy previously reported in the NEJM, the authors report this not-so-surprising result:
Among 285 patients in our cohort who also completed survey items on their expectations about chemotherapy, we found that patients with inaccurate beliefs about RT were significantly more likely to also have inaccurate beliefs about chemotherapy (P. 002 by McNemar’s test); 92% of patients with inaccurate beliefs about RT also had inaccurate beliefs about chemotherapy (Table 3).
So, basically, we have a problem in oncology. There are a lot of patients with advanced cancer who don’t understand the true nature of their prognosis. The strength of these two studies is that they looked prospectively at robust sample sizes of patients, but they both share one big inherent weakness, and that’s that neither of them can tell us why there is this huge disconnect between the patients’ perception of their prognosis and the likelihood that chemotherapy or radiation therapy could potentially cure their cancer or help them live significantly longer versus the actual prognosis. We’re missing a critical piece of information, and that’s that we don’t know what these patients were told by their oncologists and radiation oncologists regarding their actual prognosis. In both studies, the authors point out that there is a lot of evidence that physicians are often uncomfortable talking to their patients about terminal cancer and death. Although on one level, this is not surprising (after all, physicians are human beings and don’t like contemplating mortality any more than the average person—less even, perhaps, because there is the whole issue of wanting to be able to cure patients and facing a profound rebuke to their professional abilities when faced with a patient they know they can’t cure), normally one would hope that physicians would be trained to address these issues honestly, openly, and with dignity. As in everything in medicine, things tend to be messier and more complicated than that.
Of course, this goes both ways. A physician’s discomfort facing a terminally ill patient is minor compared to the anguish of that terminally ill patient himself. No one wants to face the end of his existence. No one wants to contemplate what sorts of pain, suffering, and loss of autonomy he will have to endure before the end comes. Everyone reacts with at least some denial when faced with such horrible news. Pretty much everyone wants to grasp at any straw that is within reach when faced with a terminal diagnosis. Add the patients’ tendency to deny they are dying with physician discomfort over telling them that they are, in fact, dying and that the goals of the chemotherapy and radiation therapy being proposed are strictly palliative (i.e., to address symptoms and possibly prolong life somewhat), and it is probably not so surprising that so many patients with terminal cancer misunderstand their prognosis and the likelihood that their therapy will help them in the most important way that matters to them, prolongation of life or cure of their cancer. Or, it is not so surprising that they might respond to surveys in a way that suggests that they don’t understand the direness of their prognosis, which is a different thing than necessarily misunderstanding their prognosis. Either way, it is a significant problem. As Drs. Thomas J. Smith and Dan L. Longo point out in an accompanying editorial to the NEJM study, entitled “Talking to patients about dying“, patients “also choose not to believe.” Indeed, when patients are given their actual prognosis, many will still not admit that treatment will not cure them.
As the study authors point out in both articles, an honest, accurate discussion about patient prognosis is absolutely essential. Without it, patients do not have true informed consent and can feel driven to continue toxic treatments that can reduce the quality of their remaining life without significantly increasing the likelihood that they will live longer. There is always a fine, fuzzy, grey line between continuing chemotherapy and/or radiation for palliative purposes and feeling driven to continue it at all costs because it comes to be perceived as the only hope left. In the NEJM paper, the authors write:
Should we be concerned that the majority of patients with these diseases provide responses suggesting that they do not understand that there is essentially no chance that the chemotherapy they are receiving will cure them? Chemotherapy may offer palliation and some prolongation of life, so it represents a reasonable treatment choice for some patients. However, an argument can be made that patients without a sustained understanding that chemotherapy cannot cure their cancer have not met the standard for true ongoing informed consent to their treatment.28 Furthermore, previous studies have shown that patients with advanced cancer would accept toxic treatment for even a 1% chance of cure but would be unwilling to accept the same treatment for a substantial increase in life expectancy without cure.29,30 This finding suggests that patients who do not know whether a treatment offers any possibility of cure may be compromised in their ability to make informed treatment decisions that are consonant with their preferences.29,30 Finally, and perhaps most worrisome, this misunderstanding could represent an obstacle to optimal end-of-life planning and care.
In the JCO paper, they write:
Poor understanding of treatment goals and prognosis are problematic for both medical and policy reasons. Patients cannot make well-informed decisions about their end-of-life care, taking into account risks and benefits of treatment, if they hold inaccurate beliefs about the goals of care. Studies have shown that patients with advanced cancer who do not understand and overestimate their prognosis are more likely to pursue intensive therapies near the end of life.14,28 These therapies may reduce quality of remaining life and incur substantial costs for patients with limited life expectancy. In particular, palliative RT requires daily visits for treatment, which can be burdensome for patients and families. It is possible that patients who understand that RT is not at all likely to result in cure might choose to forgo RT or pursue shorter courses of treatment. Although randomized studies suggest that short courses of RT are equally effective in many patients with poor prognosis,29 lengthier courses are more commonly given.8
In other words, this disconnect between the science-based goals of treatment and what patients perceive the goals of treatment to be, matters. It matters a lot. We exhaust a lot of money and, at least as important, precious time that terminally ill patients don’t have very much of, to administer toxic treatments that will not cure them. That does not mean that such treatments don’t palliate. When used appropriately in the correct clinical situations, radiation therapy and chemotherapy produce excellent palliation and are thus appropriate for quite a few patients with terminal cancer. However, that palliation generally means symptom reduction and maybe a slight increase in survival time.
Finally, there was an incredibly intriguing and paradoxical observation (at least to me) reported in the NEJM paper. It’s an observation that to me is relevant not just to science-based cancer care, but to the cancer quacks about which I write so often. Specifically, the authors report that patients were “less likely to provide inaccurate responses” if they reported lower scores for physician communication. That’s right, lower scores for physician communication correlate with a greater likelihood that the patient understands his prognosis and the goals of palliative chemotherapy. Specifically, in the scoring system used for patients to rate communication with their doctors, a score of 100 represents a perfect score. Using patients who scored their communication with their doctors under 80 as the baseline, the investigators calculated the odds ratio of a patient having an incorrect understanding of his prognosis to be 1.90 among those who gave a perfect score of 100 in physician communication. For patients who scored their physician’s communication between 80-99, the odds ratio was 1.32 compared to baseline. In other words, there was an inverse correlation between a patient’s rating of the communication with his physician and the likelihood that he had a correct understanding of the seriousness of his prognosis. In other words, high communication scores correlating with less understanding. Interestingly, this same correlation was not observed in the radiation therapy study reported in the JCO. In any case, the authors note:
In an era of greater measurement and accountability in health care, we need to recognize that oncologists who communicate honestly with their patients, a marker of a high quality of care, may be at risk for lower patient ratings. Our results suggest the need for targeted education to help all physicians learn to communicate honestly while also maintaining patients’ trust and regard. Efforts to incorporate earlier and more effective end-of-life care must address honestly and unambiguously patients’ unrealistic expectations about the outcomes of chemotherapy.
I think you can see the two directions where I’m going with this. First of all, as I’ve pointed out before, quality of care does not correlate with patient satisfaction scores. Companies that compile customer satisfaction ratings, like Press-Ganey, try to convince hospitals and patients that if patients are satisfied with a hospital it must mean that hospital is providing high quality care. The inherent assumption is that if patients are satisfied then the hospital and health care providers must be doing a good job. But, as I pointed out before, it’s just not that simple, and this could well be an example of that. I sometimes say that, as physicians and health care providers, there are times when we have to be “cruel to be kind in the right measure” (to steal a line from Nick Lowe). Giving a terminally ill patient a realistic assessment of his prognosis could well mean that the patient will think less highly of that physician’s communication skills—or even of that physician, period. Yet, giving the patient that assessment is the right thing to do. Sure, it’s also more complicated than that in that there are right and wrong times to deliver the news. Deliver it at the wrong time, and the patient will be less likely to accept it. Recognizing the right time to deliver such news is a physician skill that is what we call the “art” rather than the science of medicine. But sooner or later the news must be delivered.
The second direction I’m going, of course, is toward quacks and dubious cancer doctors. Almost invariably, such doctors are lionized by their patients, even the ones for whom treatment isn’t working (which is almost always nearly all of them—we are talking about quackery and non-science-based treatments here). They achieve this by giving the patient an unrealistic expectation that they can be saved, glossing over every setback as either being expected or indicative that, despite all appearances, the treatment is working after all (“the tumor will swell before it disappears” or “cyst formation means that the tumor is dying inside“). These practitioners will inevitably claim that they “never promised a cure,” and perhaps that is true if you look at what they say written on paper, but, in reality, when you take the totality of their interactions with patients into account, they do more or less promise a cure. Some even do so quite literally.
As hard as it is for many physicians and even patients to believe, patients generally welcome truthful and accurate conversations about prognosis, which do not usually increase anxiety or depression. Common reasons that physicians avoid such conversations include a number of misconceptions, some shared by patients and physicians, for which there is no evidence or for which there is actually disconfirming evidence, such as beliefs that palliative therapy will hasten the end or that it will rob the patient of hope. There’s no doubt that talking to patients about their prognosis is hard. Quite honestly, there’s a reason I’m a breast cancer surgeon instead of an oncologist, and that’s that the vast majority of my patients are potentially curable, many very curable. Yet, it’s part of our jobs. Even though I don’t come across a lot of patients with terminal disease in my practice, I want to be better at communicating with the ones I do encounter. Our patients deserve no less.