
It wasn’t that long ago that most prescriptions were custom manufactured, or compounded. A pharmacist combined raw chemicals into different dosage forms like capsules, tablets, liquids, and creams. Compounding allows complete customization of a prescription — from a new strength of a capsule, to a liquid form of a tablet, to creams with combinations of ingredients that can be completely individualized. With the proper equipment, sterile products like injectable drugs can be made in pharmacies.
These days, commercial manufacturers supply most drug products, and the need for in-pharmacy compounding has dwindled. For these products, regulators like the FDA ensure product uniformity, purity, potency, efficacy, and safety standards for consumers. You can be assured that any product approved by the FDA has gone through a barrage of manufacturing tests and multiple clinical studies. However, any retail pharmacy is still capable of providing compounded prescriptions. Some pharmacies specialize in this practice, calling themselves “compounding pharmacies” and aggressively promote the service. Organizations like the Professional Compounding Centers of America, provides training, formulations (recipes), and raw ingredients to compounding pharmacies. To pharmacies and pharmacists, compounding is a business opportunity and a way to differentiate yourself from the competition. But the enthusiasm can outpace the evidence, and there may be a lack of evidence that compounded products really are safe and effective. Now a new clinical trial raises questions about a very popular compounded product: pain creams.
While compounded pharmacy products can address important patient issues (e.g., allergy, requirement for a liquid dosage form, etc.) the fact that they undergo significantly less scrutiny than FDA-approved products has raised concern for some time. The negligence that occurred at the New England Compounding Center killed 64 people from fungal meningitis that was a consequence of shoddy manufacturing standards. Evidence standards can also be questionable, as I’ve pointed out before, where compounding pharmacies have a symbiotic relationship with advocates for practices like “chelation”, used in circumstances where the benefits are unproven.
Treating chronic pain
Chronic pain is the most common cause of long-term disability and can occur as a consequence of diseases like cancer. Despite the societal burden of pain, there are few truly effective (and risk-free) medical options. Opioids are often used but with mixed results and legitimate concerns about addiction. Non-steroidal anti-inflammatory drugs (NSAIDs) can be effective for some forms of pain, but their side-effect profile is considerable. I’ve written before about topical NSAIDs which despite my initial skepticism do have some evidence of effectiveness for osteoarthritis and acute pain and fewer side effects compared to NSAID pills.
Given the positive clinical trials from commercially-manufactured products like Voltaren cream, and recognizing the limitations of other treatment options for chronic pain, it is perhaps no surprise that there has been growing interest in preparing other products for topical use for the treatment of pain. Compounding pharmacies will manufacture multiple products marketed for the treatment of pain. Muscle relaxants or drugs like ketamine and lidocaine can be prepared for topical use. Sometimes multiple ingredients are compounded into a single cream. In the paper we’re about to discuss, the authors, who are affiliated with a US military treatment facility, note that the Department of Defense’s health system drug program was spending $6 million per day on compounded medications in 2015, an amount that has been growing over time. Soaring expenditures and a lack of efficacy data led to the current study, which sought to examine if these expensive, customized products actually work.
The study
This study is from Robert E. Brutcher and colleagues at the Walter Reed National Military Medical Center, entitled “Compounded Topical Pain Creams to Treat Localized Chronic Pain“, published in the Annals of Internal Medicine in February, 2019.This was a double-blind, randomized, parallel study of 399 patients that compared three different compounded pain products versus placebo. Participants were from an academic military treatment facility and had to be 18-90 years old, with localized pain on a pain scale of 4 or more on a 1 to 10 scale, that had lasted more than 6 weeks. More than 40% were active duty personnel. Patients were randomized to one of three products (active or placebo) based on the type of pain they were experiencing and the assessment of the treating physician.
- Nociceptive pain (pain typically caused by injury or inflammation): The active treatment was 10% ketoprofen, 2% baclofen, 2% cyclobenzaprine and 2% lidocaine.
- Neuropathic pain (pain due to nerve injury): The active treatment was 10% ketamine, 6% gabapentin, 0.2% clonidine, and 2% lidocaine.
- Mixed pain: The active treatment was 10% ketamine, 6% gabapentin, 3% diclofenac, 2% baclofen, 2% cyclobenzaprine, and 2% lidocaine.
Creams were compounded in a typical pharmacy compounding base (carrier). The concentrations of active ingredients were based on previous trials and commonly marketed compounded formulas. Creams were applied to the affected area 3 times per day.
The primary outcome of interest was the average pain score one month after treatment. A positive response was deemed to be a reduction in pain score of 2 (scale of 1-10) and a score above 3 on a 5-point satisfaction scale. There were a number of secondary outcomes, and patients with positive-reported outcomes were followed for 3 full months.
The results
The (compounded) drugs don’t work. All patients improved slightly, but the change in pain score at 1 month, and at 3 months did not differ significantly between the drug and placebo for all three pain groups. There were also no differences in functional improvement or satisfaction with the product. The placebo was as effective as the “active” treatment.
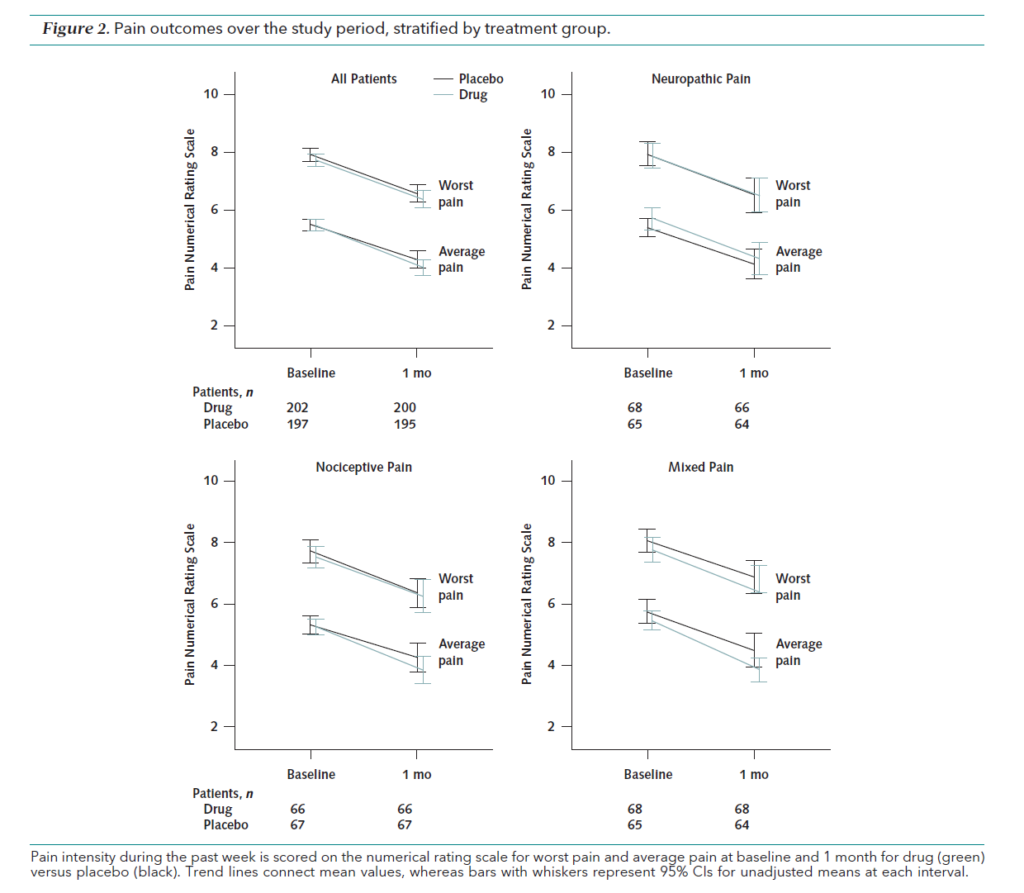
Both the active treatments and the placebos were generally well tolerated and there were no differences overall in side effects, with the exception of slightly more skin irritation reported in the active treatment group.
The implications
This study found no evidence that 3 different compounded pain creams have any effect on different forms of pain. This study is consistent with some other research but is also at odds with some randomized trials that have shown effectiveness for products like topical NSAIDs for conditions like knee osteoarthritis. Limitations of this study include the variety of medical conditions included, which might miss effectiveness in a specific type or location of pain (e.g., knee osteoarthritis). Overall, this trial gives important information that should lead health professionals and consumers to question the effectiveness of compounded pain creams.

