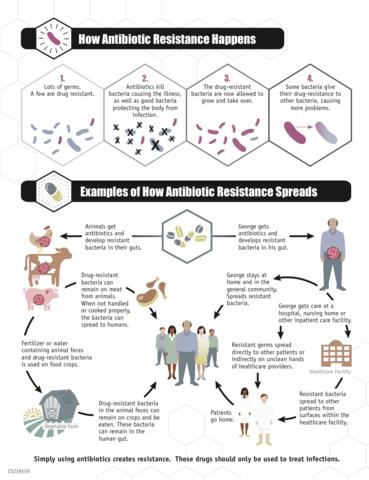
Editor’s note: Today we present a guest post from fourth-year medical student Joshua Horton, about the looming problem of antibiotic resistance. Welcome!
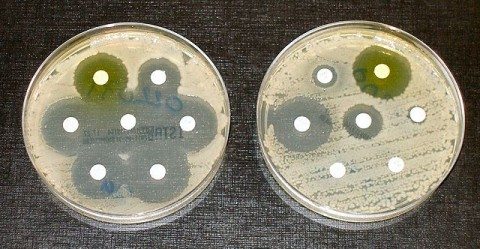
Antibiotic sensitive (left) and resistant (right) bacterial colonies. They probably shouldn’t be that close together.
I read a study recently that alarmed me: acute bronchitis is a condition that rarely requires antibiotics, but three quarters of patients presenting with this condition receive a prescription for antibiotics. Even more worrisome, this statistic has not changed in 20 years. To those of us on the cusp of a career in medicine – I am a fourth year medical student – this is terrifying because we are going to have to deal with the consequences. Antibiotic overuse leads to:
- Increased morbidity and mortality for patients infected with resistant bugs
- Unnecessary and exorbitant healthcare expenditures
- The potential to cultivate multi-resistant bacteria that could spread to pandemic proportions
We still take them for granted, but antibiotics are a finite resource. Each time we breed a bug that is resistant to a particular drug, we are forced to relinquish that gun from our armamentarium. With fiscal impetus for pharma to develop new antibiotics waning, we may have reached a point of no return. That’s why this study scared me. Hopefully, clever techniques based on behavioral theory designed to reduce antibiotic prescribing may pull us back from that edge – read on to learn more.
We have known about the problem of antibiotic overuse and how to control it for a while, but the response has been slow. Experts, including Otto Cars (sweet name, man) of Uppsala University in Sweden, believe that the lack of morbidity and mortality data associated with antibiotic resistance prevents politicians and public health workers from reacting robustly. We all know how efficient and reliable political and public policy change can be. In the meantime, what about the ground level? Antibiotic resistance can be tied to the misuse and overuse of antibiotics by both patients and physicians. What are we doing wrong?
The physician and antibiotic resistance
According to a recent systematic review, the majority of physicians have “heard about antibiotic resistance, believe it is a serious problem, believe that using too many antibiotics is the main cause, and know some of the evidence-based strategies to reduce it.” Sounds great, right? Unfortunately, most deflect responsibility.
More physicians attributed resistance to a being a “global problem,” due to patient factors, or emanating from other countries or healthcare settings than said it was a problem for their own practice. Further, they considered resistance to be a “low priority and a distant consequence of antibiotic prescribing.” (I have to be honest: this was also my opinion before getting into the later years of medical school and becoming familiar with the literature.) And when patients insist on antibiotics in situations when they are not indicated, physicians will often relent under this social pressure for fear of dissatisfying or even losing the patient. I would hope this could be a prime opportunity to educate the patient, rather than caving to the pressure.
For now, doctors are (still) prescribing far too many antibiotics.
The patient and antibiotic resistance
Another review, this time on patient perceptions of antibiotic resistance, compiled studies from Europe, Asia, and North America. If you are familiar with this topic or sniffed out the theme, you can predict the next part. While most patients had heard of antibiotic resistance (some even knew strategies to reduce it), they “largely attributed its development to the actions of others” and thought changes in physician behavior were the key to its reduction.
There is a culture of “not my problem.” But I think with that mindset, soon enough it will be everyone’s problem.
Humans are not inherently altruistic, at least not to a significant degree (except maybe to their kin – Richard Dawkins has some profound thoughts on this, check out chapter 6 of The Selfish Gene). It is, therefore, unrealistic to expect a patient to “suffer through an illness” sans treatment simply for the ideological goal of preventing global resistance, when (in their mind) antibiotics would cure them. It may work better to educate the patient regarding the risk of becoming a long-term carrier of resistant bugs and the risk to themselves of more severe infection with these bugs.
Despite this talk of physician- and patient-specific misconceptions and failures regarding antibiotic use, much of the problem arises outside of this doctor-patient contract. At home, patients are their own doctors and the opportunity for education and intervention is lost. This is where much of the consumer-created damage leading to antibiotic resistance occurs. Patients feel better and stop their antibiotic course halfway through. Patients suffering from a simple common cold prescribe themselves their sister-in-law’s expired Z-Pak. Patients take expired drugs or reduced doses.
There is a significant knowledge barrier between patients and the responsible use of antibiotics. And that barrier is larger in some places than others.
The world and antibiotic resistance
We are only as strong as our weakest link. It’s important to study public knowledge of antibiotic resistance in developed countries with healthcare systems, but the ultimate multi-resistant bug could come from anywhere in the world.
In November, the World Health Organization (WHO) published findings of a large global survey assessing the understanding of antibiotic resistance among subsets of the world population. Twelve countries were surveyed, chosen to encompass varying income levels, populations, and preexisting knowledge of antibiotic resistance (Nigeria, South Africa, Barbados, Mexico, India, Indonesia, Egypt, Sudan, Russian Federation, Serbia, China and Vietnam). Surveys were conducted online or face-to-face and nearly 10,000 (n=9,772) response sets were recorded. The key findings, as listed in the report, were:
- 65% of respondents had taken antibiotics within the last six months
- Antibiotic use was higher in poorer countries
- 72% acknowledge that many infections are becoming resistant to antibiotics
- 25% think it’s okay to use antibiotics from a friend or family member
- 43% think it’s okay to re-use antibiotics that helped them in the past
- 32% say they should stop taking antibiotics when they feel better, rather than finish the course (especially in Sudan, Egypt, and China)
- 64% reported that cold and flu should be treated with antibiotics
- 57% think there is nothing people like them can do to help prevent resistance
- 76% believe “resistance” means that their body is becoming resistant to the drug
Obviously there is much work to be done! As in the other studies quoted above, most people can identify that antibiotic resistance is a problem and are aware of some basic prevention (e.g. hand-washing). However, there’s not much understanding around the world of any more than that: what resistance actually is, how it develops, and what can be done to prevent it. Echoing many of the studies coming before, this report makes some general recommendations to develop a “better understanding of why antibiotics are being used without having been prescribed” and to increase public education about “which conditions can be treated with antibiotics, why we should limit intake of antibiotics, and the importance of finishing the prescribed course.”
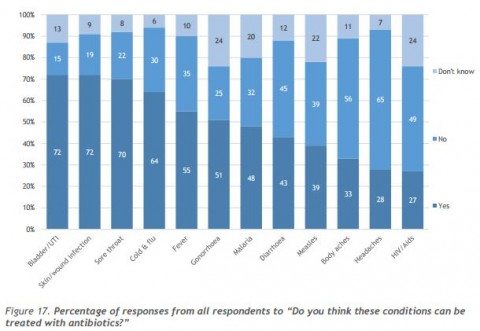
Figure 17 from the WHO report: % respondents and the conditions they believed to be treatable with antibiotics (click to embiggen)
A significant limitation of this report was the fact that only two countries per WHO region were included, therefore it was not a supremely representative sample of the individual regions or the global population. Closed-ended questions were also used in the survey, which allowed for only a limited knowledge of the patient’s actual understanding. Finally, as with all survey-based studies, there is a risk of response bias (potentially social desirability bias).
The official conclusions
Through the implementation of evidence-based approaches to public education and proper antibiotic use, there is significant potential for practice improvement. According to the WHO report, physicians should be frugal with antibiotic prescription, become familiar with resistance patterns, and take care to prescribe only drugs indicated for the suspected bug.
Patients, in addition to employing proper hygiene and infection-prevention measures, should follow antibiotic dosing and course instructions precisely, never sharing antibiotics or stopping a course early (even if they are feeling better).
From a systems perspective, improvements in resistance tracking and research into new diagnostic tools and medications are sorely needed.
Is that all?
The next steps
I am finding my fourth year of medical school to be an interesting time. About to begin my training in my chosen specialty within medicine – with all of the required (and graded) courses behind me – I am struggling to strike a balance between the diligent, hard-working version of myself that got me through the difficult years of medical school and the happy-go-lucky version of myself that doesn’t want to be on campus unless absolutely necessary. It’s the medical school version of the “senioritis” people describe feeling in their last semester of high school or college.
However, it was in one of my recent fits of diligence (while reviewing the literature for an unrelated project) that I came across the study I kicked off this post with. It would have been easy to stop there (especially with my self-motivation at an all-time low), but something about the topic really got under my skin. So, doctors should prescribe fewer antibiotics and patients should use them as prescribed. Great. But…um…what is actually being done to create these changes? Anything? It doesn’t seem there’s been much progress. And I’m going to be a doctor soon, so can we get our asses in gear?
Behavioral modification
Lucky for me, while working on revisions for this piece, the latest issue of the Journal of the American Medical Association (JAMA) landed in my inbox. In it, a report by researchers from Harvard and UCLA details the findings of a study comparing various behavioral modification techniques for inappropriate antibiotic prescribing by primary care physicians. Physicians who wrote an prescription for antibiotics to treat an acute respiratory infections encountered one of three prompts while updating their patient’s electronic medical record:
- Suggested alternatives: upon signing the prescription, a “pop-up” appeared within the electronic medical record with a statement along the lines of, “antibiotics are not generally recommended for this condition” and alternative treatment strategies were suggested.
- Accountable justification: upon signing the prescription, the physician would be required to enter a free-text justification for writing the prescription, which would be recorded in the patient’s chart.
- Peer comparison (my money is on this one; most doctors are, after all, competitive creatures at their core): physicians received an email message informing them that they were a “top performer” (in the top decile of appropriate antibiotic use) or “not a top performer” (if they found themselves in the bottom decile).
Forty-seven primary care practices in Boston and Los Angeles comprising 248 physicians were randomized to encounter various combinations of these interventions and then their prescription patterns were followed for 18 months. The first interesting finding from this study was that the negative controls (those encountering none of the interventions) decreased their antibiotic use by 11%. As the authors point out, this is likely due to the Hawthorne effect: the tendency to change behaviors while participating in a study just due to the fact that one knows their actions are being scrutinized. I bring this up mostly to illustrate the importance of being judicious when reading scientific evidence, even the most well-controlled studies in the highest-impact journals. Most SBM readers will be aware of this, but extraneous effects such as these are rampant in scientific research and the implications must be considered. In this case, the Hawthorne effect is not necessarily a limitation, but it may have dampened the effect size of the interventions.
These were the results, in terms of rate of inappropriate antibiotic prescription:
- Suggested alternatives: 22.1% pre-intervention to 6.1% post-intervention (16% drop)
- Accountable justification: 23.2% to 5.2 % (18% drop)
- Peer comparison: 19.9% to 3.7% (16.2% drop)
Only the drops observed in strategies #2 and #3 were found to be statistically significant compared to the amount that the control group rates reduced. No combination of techniques was better or worse than any single technique. Importantly, an increase in complications or return visits stemming from reduced antibiotic prescription for acute respiratory infection was not observed – reducing antibiotic prescriptions did not seem to result in worse outcomes.
As Jeffrey Gerber alludes to in his editorial discussion of this study in the same issue of JAMA, these interventions have two major strengths. First, while antibiotic stewardship programs are now common in hospitals, antibiotic usage trends are not currently monitored in the outpatient setting. This study did just that. The methods employed by the researchers could serve as a model for future tracking systems in the outpatient setting. Second, and more immediately salient, the interventions are simple: they do not create significant waste (time, money, or resources) within the system and could be easily implemented across all electronic medical record systems.
Of course, the study was limited in a couple areas: namely that the sample sizes were relatively low and the physician practices received monetary compensation for their participation, which Dr. Gerber believes may limit the generalizability of the results.
One more interesting finding of the study: the only intervention that was not found to be statistically significant (#1, suggested alternatives) was also the only one that did not involve a social component. Just food for thought.
This has not been an exhaustive account of the literature on this topic, just some recent, relevant data to pique your interest and hopefully light a fire under those of you in a position to incite change. As I write this final paragraph, I am on a flight to my final residency interview of what has been a grueling interview season (at one point, I had six interviews in seven days in cities scattered across the country). Thinking about transitioning from this to the next step in my training, I also think about what the landscape of antibiotic usage and resistance will look like in my practice in the decades to come. I wanted to become a doctor to help people, and at the beginning of my first year of medical school, I pledged an oath to “Do No Harm.” But we are doing harm. Giving a patient an antibiotic for a viral sinus infection in order to pacify them today is putting the patient sitting across from me in my office 20 years from now in harm’s way. What am I going to give them when we have used up all of our antibiotic options or when they have a bug that is resistant to even our biggest guns? We know that change is needed and, increasingly, we are developing the tools to enact that change. So what are we waiting for?
About the author

Joshua Horton is a graduating 4th year medical student at New York University School of Medicine who was born and raised in Indiana and completed his undergraduate studies in biochemistry at Indiana University-Purdue University Indianapolis. He is heading to the Medical University of South Carolina next year to start a residency in Otolaryngology-Head and Neck Surgery. In addition to seeing patients and operating, he hopes to make clinical research and student/resident mentoring a significant component of his career.