If science-based medicine is unaffordable, then your care won’t be science-based. Prescription drug costs are one of the biggest concerns in health care today. There seems to be no upper limit on prices, with some new treatments costing over $1,000 per day. The arrival of new drugs to treat (and cure) hepatitis C has created a perfect pharmaceutical storm: highly effective treatments, a large population of potential patients, and huge per-patient costs. It’s renewing the debate about whether important medical treatments are being priced out of the reach of the patients that need them. It’s not just hepatitis. Cancer drug costs are rising as well, driven by more patients and new drugs that in some cases are transforming our expectations about what cancer drugs can do. And while many of us rely on some form of drug insurance to protect us from high drug costs, insurers are struggling with balancing coverage and premiums: A report by Express Scripts paints a grim picture:
An estimated 576,000 Americans spent more than the median household income on prescription medications in 2014. This population of patients grew an astounding 63% from 2013. Further, the population of patients with costs of $100,000 or more nearly tripled during the same time period, to nearly 140,000 people. The total cost impact to payers from both patient populations is an unsustainable $52 billion a year.
This isn’t just an issue in the United States. Prescription drug costs are climbing around the world, because we’re effectively all in this together: We all rely on private companies to bring new drugs to market, and we’re largely buying the same drugs from the same small group of companies. Because ready access to safe and effective prescription drugs is so important to the practice of medicine and the delivery of health care, the pharmaceutical industry is heavily regulated – not just by the FDA, but by regulators worldwide. Yet despite the dual requirements of regulatory disclosure and the financial obligation to be transparent (as many pharmaceutical companies are publicly-held), little is known about how much it costs to bring drugs to market, and how manufacturers arrive at their selling prices. Pharmaceutical manufacturers claim that high drug costs reflect the high costs of research and development (R&D), and provide the incentives for companies to invest heavily and take risks, when many drugs may never make it to market. Are they correct?
I’ve written before about different attempts to estimate how much it costs to develop a drug. This is obvious a point of contention, with pharmaceutical companies interested in profiling the risk and significant investments they make, while using high prices as the justification to support continued risky R&D. Detractors and critics believe that these estimates are overstated by industry. The most well-known analysis of drug development costs is by Joe DiMasi at the Tufts Center for the Study of Drug Development. DiMasi collected R&D data for a basket of drugs that were volunteered by pharmaceutical companies, in order to come up with average drug development estimates. His original calculation was $802 million per drug, back in 2003. Today his updated estimate is $2.6 billion per drug. But there have been multiple critiques of DiMasi’s methodology and estimates over the years. Even Andrew Witty, the head of GlaxoSmithKine, one of the largest pharmaceutical companies, has called the $1 billion price tag “a myth” of industry. A critique of the latest DiMasi cost estimate appeared the New England Journal of Medicine earlier this month. In a commentary, Jerry Avorn made the following observations:
- The raw data from DiMasi is not available for analysis (and likely never will be, given it was provided in confidence by manufacturers).
- The analysis assumes 80% of new compounds are abandoned during development (and costs them accordingly) yet this number cannot be independently verified.
- $1.2 billion of the $2.6 billion is the “cost of capital”, rather than a direct cost of development. The cost of capital is estimated at 10.6% which seem high compared to rates pharmaceutical companies pay on their own bonds. Given the significant cash holdings by the bigger companies, access to capital does not seem to be an issue.
- Public subsidies into drug development are not addressed or accounted for.
Avorn highlights that not all costs are climbing: FDA approval times are shortening (compared to prior analyses), reducing the overall estimated costs of development. Given the collective costs of the “failures” are higher than the costs of the successful drugs, the DiMasi analysis suggests that it is the development process itself, rather than regulation, that may be driving the pricing of drugs. Avorn concludes with a call for more transparency on development costs as a first step in a dialogue on how to support research, while appropriately rewarding innovation. Some American state legislators aren’t waiting for industry reaction – and they’re proposing legislation to force the discussion.
Forcing R&D cost disclosure
Responding to repeated calls for transparency, and struggling with their own prescription drug cost bills, some states are looking to legislation to help manage prescription drug costs, perhaps by publicly pressuring manufacturers into reconsidering their pricing. The latest strategy was announced recently by New York Senator Ruben Diaz:
New York is the latest state to introduce a “pharmaceutical cost transparency act,” following five others—California, Oregon, Massachusetts, North Carolina, and Pennsylvania. Like the bills before it, New York would require pharmaceutical manufacturers to submit a report to the state outlining the total costs of the production of certain expensive drugs, with the information to be published on a public website.
The bill would force pharmaceutical manufacturers to disclose specific information for a drug that costs more than $10,000 per year – a detailed disclosure of production and sales data:
- Total R&D paid by the manufacturer (and any predecessor)
- The total costs of clinical trials and regulatory costs
- Total costs to manufacture (fabricate) the drug product itself
- Total costs paid by other entities (e.g., governments) on direct R&D
- Any other costs to acquire the drugs (e.g., patents and licensing)
- Total marketing and advertising costs, including costs of promotion to physicians
- The average wholesale price, with changes over time
- Total profit attributable to the drug
- Financial assistance provided by manufacturers
While this sounds like it might be an attractive approach to forcing transparency, it ignores a very real cost that the DiMasi analysis acknowledges: Most new drugs don’t succeed, and companies depend on the “hits” to stay profitable. Looking only at the cost of the “hits” ignore much of the real cost of drug development. Hundreds of millions of dollars, if not billions, have been invested in studying drugs to treat Alzheimer’s disease with strikeout after strikeout. Pfizer had invested about $800 million in torcetrapib when it discovered the drug was killing patients instead of preventing cardiovascular disease. The road to blockbuster drugs, like the new hepatitis treatments, is paved in part by well-documented, very costly, pharmaceutical development failures.
Even if we acknowledge (and I think we must) that companies have to cover the costs of their failures as well as their hits, is there any value in forcing disclosure about the hits alone? This approach is almost certainly going to underestimate the “real” cost of development, as not every cost can be traced to a specific drug. DiMasi estimated the costs of some of the earliest development stages of drug development, recognizing that you can’t attribute costs to any specific chemical. How else can you allocate the costs of pre-clinical research that may generate dozens or even hundreds of possible drug candidates? The development stage includes screening promising chemicals, validating them, and then modifying the chemical structure repeatedly to find the compound that represents the best possible candidate for future study. Only a fraction of drugs ever proceed beyond this step. Derek Lowe goes over this in much more detail in one of his old posts – he’s in the business of drug development, and knows of what he speaks. This is a tedious, time-consuming, often unproductive process.
There are also the ongoing costs of making a drug available – the costs of meeting regulatory requirements, and the overhead cost of running a business with tens of thousands of employees. The legislation ignores all of those costs as well, focusing only on that can be directly linked to a chemical. If this type of legislation succeeds, while it may be more transparent (in part) than the DiMasi paper, it will be no more helpful in helping us understand pharmaceutical R&D costs and pricing.
Another approach to estimating costs
Matthew Herper writing in Forbes in 2013, took a crude yet simple approach to estimating costs. He added up the R&D expenses disclosed by pharmaceutical companies on their financial statements and divided it by the number of drugs produced over time. What he concluded was that some companies were spending astonishing amounts to produce new drugs. Abbott (now called AbbVie) was spending $13 billion per drug (and device) produced. Sanofi appeared to be spending $10 billion per drug. And so on, down to small companies which appear to be bringing new products to market for well under $100 million. Herper noted that 66 companies launched only one drug in decade, with a median cost of $350 million for that single product. Herper’s analysis adds some credibility to the high estimates from Tufts, and only seems to suggest how unproductive the R&D process appears to be for the bigger companies. But his analysis misses a lot of context: It doesn’t adjust for new uses for old drugs. Nor does it clarify the effects of mergers and acquisitions on costs. And while these are real limitations, the analysis looks like a reasonable approximation of the challenge. As I’ve noted before, while there have been some remarkable new and effective drugs developed, the industry as a whole appears to be suffering from a productivity problem, as illustrated by this 2012 image from a paper by Scannell et al. (now paywalled), showing the number of drugs produced per billion of dollars in R&D:
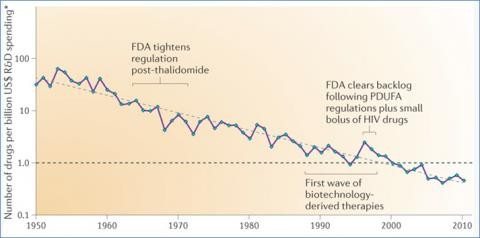
From Scannell et al. (click to embiggen)
Will breaking patents help?
Companies can only maintain high prices in the market when they have patent protection. Another approach that’s been proposed to deal with high prices has been to allow patents on high-cost drugs to be broken, to allow early generic competition. Driven in part by skepticism about development costs, advocates for this approach argue that drug costs that bankrupt individuals are immoral and unethical. Better to change the rules, they argue, rather than let patient suffer. And this kind of proposal isn’t restricted to poor countries. Senator Bernie Sanders has proposed the same approach to help the Department of Veterans Affairs afford the cost of the new hepatitis drugs:
Our nation’s veterans cannot, and should not, be denied treatment while drug companies rake in billions of dollars in profits.
In some cases, pharmaceutical companies are breaking their own patents. Gilead is the patent-holder and manufacturer of the hepatitis blockbuster sofosbuvir (Sovaldi) and it is allowing generic companies to produce and sell their product into developing countries. Realistically, I don’t see arbitrary patent-breaking as gaining much traction, as most realize that it would possibly do more to undermine R&D investment in new drugs than any other strategy. Changing the rules of the game, when drug development can take decades, could jeopardize any investment in the areas where we need new treatments, particularly where the rewards seem modest at best. Antibiotics are an example of the disconnect between medical need (high) and drugs in development (few). Growing concerns about antibiotic resistance are heightened by the reality that there are few new antibiotics in development. If we don’t have effective antibiotics, even everyday infections can be deadly. What seems necessary, perhaps is a better linkage between the value of new treatments and the incentives that in place to develop them. Breaking the patents on drugs we don’t want to pay for wouldn’t address this problem, or the overall productivity challenge in development.
Is “cost of development” even the right question to ask?
To continue to improve science-based medicine, we need to find a way to introduce new innovative treatments at prices that that are affordable. Looking at R&D costs for solutions may be the wrong question to ask. What really matters is if a particular treatment delivers value. A new treatment may be worth the high cost if it avoids other costs, such as the potential for a vaccine to prevent cancer (e.g., HPV) or for a drug to eliminate the need for a liver transplant (e.g., the new hepatitis treatments). From that perspective, the R&D costs aren’t something we should focus on. We don’t ask (or care) what it costs to develop a new car, or the latest phone. What matters is whether we value the benefits of the “new” versus the status quo. The challenge with drugs (and with healthcare in general) is that (1) assessing value is difficult when it comes to health treatments and (2) we are often shielded (via insurance) from directly bearing the costs ourselves. New drugs need to show value in the calculation of their cost. And we also need to do what we can to bring the costs of development (and innovation) down. Otherwise we run the risk of less innovation and fewer new treatments that don’t offer value for the price.
Conclusion
As health professionals, consumers and as patients, it’s perhaps relevant to question what it is we expect in terms of new medicine. We’ve become accustomed to new treatments that transform diseases or even prevent them. Yet productivity is dropping and costs are rising. Is there a common ground we can agree on? There would likely be a strong consensus that we want new treatments that are truly innovative (safer and more effective) compared to current treatments. And these innovations have to be valued by patients as delivering meaningful benefits. To get there, we can likely all agree that we must support measures that foster R&D, particularly those that help identify promising drug candidates sooner, thereby reducing the costs of failures. We need to balance rigorous regulation during development with the imperative to ensure patient safety during trials, and after the drug is released. And we cannot overlook the need to understand how much better a new treatment is compared to what we’re doing now. In doing so, we should get a better sense of what it should cost to develop a new drug. And the price we eventually pay needs to support a R&D culture that continues to advance, in an affordable way, improvements in the way we deliver medical care today, and into the future.
Photo via flickr user Stockmonkeys.com via a CC licence.
